Erradicación del Helicobacter pylori para la prevención del cáncer gástrico
Appendices
Appendix 1. Glossary of terms
Antibiotics: amoxicillin, clarithromycin, macrolide, 5‐nitroimidazole
Antrum: the bottom section of the stomach
Ascorbic acid: vitamin C
Asymptomatic: without symptoms
Bismuth: a medication that reduces stomach acid
Carbon‐urea breath test: a diagnostic test that uses an isotope of carbon to detect H. pylori
Carcinogenic: causing cancer
Chronic: long term
Dyspepsia: indigestion
Endoscopy: a diagnostic test using a telescope to look into the stomach and intestines
Gastric atrophy: loss of the normal glands in the stomach
Gastric metaplasia: a change in the cells in the stomach from one type to another
Gastric: stomach
Gastritis: inflammation of the stomach
Gastrointestinal: involving the stomach and intestines
Histamine2‐receptor antagonist: a medication that reduces stomach acid
Histology: the appearance of biopsies from an organ under the microscope
Luminal: in the cavity of a hollow organ, such as the stomach or intestine
Malignant: cancerous
Mortality: death
Mucosa: in the lining of a hollow organ, such as the stomach or intestine
Neoplasia: abnormal growth of tissue, often cancerous
Nested case‐control study: variation of a case‐control study in which only a subset of controls from the cohort are compared to the cases
Non‐cardia: involving the antrum or body of the stomach
Occult: unobvious or hidden
Proton pump inhibitor: a medication that reduces stomach acid
Ranitidine bismuth citrate: a medication that reduces stomach acid
Rapid urease testing: a biopsy test from the mucosa of the stomach, which is placed in a special liquid, to detect H. pylori
Serology: a blood test to detect antibodies against an infection such as H. pylori
Appendix 2. CENTRAL search strategy
-
MeSH descriptor: [Helicobacter] explode all trees
-
MeSH descriptor: [Helicobacter Infections] explode all trees
-
MeSH descriptor: [Helicobacter pylori] explode all trees
-
helicobacter or pylori or pyloridis or Campylobacter (Word variations have been searched)
-
#1 OR #2 OR #3 OR #4
-
MeSH descriptor: [Stomach Neoplasms] explode all trees
-
MeSH descriptor: [Lymphoma, B‐Cell, Marginal Zone] explode all trees
-
mucosa associated lymphoid tissue lymphoma or MALT (Word variations have been searched)
-
((stomach or gastric) near/3 (cancer* or carcinoma* or neoplas* or tumor* or tumour* or lymphoma or adenocarcinoma or malignant)) (Word variations have been searched)
-
#6 or #7 or #8 or #9
-
#5 and #10
-
MeSH descriptor: [Proton Pump Inhibitors] explode all trees
-
MeSH descriptor: [Omeprazole] explode all trees
-
MeSH descriptor: [Esomeprazole Sodium] explode all trees
-
proton pump inhibitor* or PPI* (Word variations have been searched)
-
omeprazole or losec or nexium or prilosec or rapinex or zegerid or ocid or Lomac or Omepral or Omez (Word variations have been searched)
-
pantoprazole or protium or protonix or Pantotab or Pantopan or Pantozol or Pantor or Pantoloc or Astropan or Controloc or Pantecta or Inipomp or Somac or Pantodac or Zurcal or Zentro (Word variations have been searched)
-
lansoprazole or lanzoprazole or ag 1749 or agopton or bamalite or lanzor or monolitum or ogast or ogastro or opiren or prevacid or prezal or pro ulco or promeco or takepron or ulpax or zoton (Word variations have been searched)
-
esomeprazole or nexium (Word variations have been searched)
-
dexlansoprazole or Kapidex or Dexilant or tenatoprazole or CAS 113712‐98‐4 or STU‐Na (Word variations have been searched)
-
#12 or #13 or #14 or #15 or #16 or #17 or #18 or #19 or #20
-
MeSH descriptor: [Histamine H2 Antagonists] explode all trees
-
MeSH descriptor: [Cimetidine] explode all trees
-
MeSH descriptor: [Ranitidine] explode all trees
-
MeSH descriptor: [Famotidine] explode all trees
-
MeSH descriptor: [Nizatidine] explode all trees
-
H2 receptor antagonist* or H2RA* or H2‐RA* (Word variations have been searched)
-
ranitidine or zantac or famotidine or pepcid or cimetidine or tagamet or nizatidine or Axid (Word variations have been searched)
-
histamine2 receptor antagonist* (Word variations have been searched)
-
#22 or #23 or #24 or #25 or #26 or #27 or #28 or #29
-
MeSH descriptor: [Bismuth] explode all trees
-
MeSH descriptor: [Amoxicillin] explode all trees
-
MeSH descriptor: [Clarithromycin] explode all trees
-
MeSH descriptor: [Nitroimidazoles] explode all trees
-
MeSH descriptor: [Macrolides] explode all trees
-
metronidazole or tinidazole or amoxicillin or amoxycillin (Word variations have been searched)
-
clarithromycin or azithromycin or roxithromycin (Word variations have been searched)
-
bismuth or nitroimidazole* or macrolide* (Word variations have been searched)
-
#31 or #32 or #33 or #34 or #35 or #36 or #37 or #38
-
#21 or #30 or #39
-
#11 and #40
Appendix 3. MEDLINE search strategy
-
exp Helicobacter/ or exp Helicobacter pylori/ or exp Helicobacter Infections/
-
(helicobacter or campylobacter).mp.
-
(pylori or pyloridis or HP).ti,ab,kw.
-
or/1‐3
-
exp Stomach Neoplasms/
-
exp Lymphoma, B‐Cell, Marginal Zone/
-
(mucosa associated lymphoid tissue lymphoma or MALT).ti,ab,kw.
-
((stomach or gastric) adj3 (cancer* or carcinoma* or neoplas* or tumor* or tumour* or lymphoma or adenocarcinoma or malignant)).ti,ab,kw.
-
or/5‐8
-
4 and 9
-
randomized controlled trial.pt.
-
controlled clinical trial.pt.
-
placebo.ab.
-
drug therapy.fs.
-
random$.mp.
-
trial.ab.
-
groups.ab.
-
or/11‐17
-
case report/
-
Case study/
-
exp animals/ not humans/
-
or/19‐21
-
18 not 22
-
10 and 23
-
exp Omeprazole/ or exp Proton Pump Inhibitors/
-
exp esomeprazole/ or exp Esomeprazole Sodium/
-
((proton adj2 pump adj2 inhibitor$) or PPI or PPIs).ti,ab,kw.
-
(omeprazole or losec or nexium or prilosec or rapinex or zegerid or ocid or Lomac or Omepral or Omez).tw.
-
(pantoprazole or protium or protonix or Pantotab or Pantopan or Pantozol or Pantor or Pantoloc or Astropan or Controloc or Pantecta or Inipomp or Somac or Pantodac or Zurcal or Zentro).tw.
-
(lansoprazole or lanzoprazole or ag 1749 or agopton or bamalite or lanzor or monolitum or ogast or ogastro or opiren or prevacid or prezal or pro ulco or promeco or takepron or ulpax or zoton).tw.
-
(rabeprazole or aciphex or dexrabeprazole or e 3810 or ly‐307640 or pariet).tw.
-
(esomeprazole or nexium).tw.
-
(dexlansoprazole or Kapidex or Dexilant).tw.
-
(tenatoprazole or CAS 113712‐98‐4 or STU‐Na).tw.
-
exp lansoprazole or exp rabeprazole/
-
or/25‐35
-
exp Histamine H2 Antagonists/
-
exp Cimetidine/
-
exp Ranitidine/
-
exp Famotidine/
-
exp Nizatidine/
-
(H2 receptor antagonist* or H2RA* or H2‐RA).tw.
-
(ranitidine or zantac or famotidine or pepcid or cimetidine or tagamet or nizatidine or Axid).tw.
-
histamine2 receptor antagonist.tw.
-
or/37‐44
-
exp Bismuth/
-
exp Amoxicillin/
-
exp Clarithromycin/
-
exp Nitroimidazoles/
-
exp Macrolides/
-
(metronidazole or tinidazole or amoxicillin or amoxycillin).ti,ab,kw.
-
(clarithromycin or azithromycin or roxithromycin).ti,ab,kw.
-
(bismuth or nitroimidazole* or macrolide*).ti,ab,kw.
-
or/37‐53
-
36 or 45 or 54
-
24 and 55
Appendix 4. EMBASE search strategy
-
exp Helicobacter infection/ or exp Helicobacter/ or exp Helicobacter pylori/
-
(helicobacter or campylobacter).mp.
-
(pylori or pyloridis or HP).ti,ab.
-
or/1‐3
-
exp stomach cancer/ or exp stomach tumor/
-
exp stomach adenocarcinoma/
-
exp stomach lymphoma/
-
exp mucosa associated lymphoid tissue lymphoma/
-
(mucosa associated lymphoid tissue lymphoma or MALT).ti,ab,kw.
-
((stomach or gastric) adj3 (cancer* or carcinoma* or neoplasia or neoplasm* or neoplastic or tumor* or tumour* or lymphoma or adenocarcinoma or malignant)).ti,ab,kw.
-
or/5‐10
-
4 and 11
-
(random$ or clinical trial$ or blind$ or placebo$).mp.
-
exp health care quality/
-
exp randomized controlled trial/
-
13 or 14 or 15
-
Case study/
-
exp animal/ not exp human/
-
case report/
-
17 or 18 or 19
-
16 not 20
-
12 and 21
-
exp omeprazole/ or exp esomeprazole/ or exp proton pump inhibitor/ or exp rabeprazole/ or exp lansoprazole/ or exp pantoprazole/
-
((proton adj2 pump adj2 inhibitor$) or PPI or PPIs).ti,ab,kw.
-
(omeprazole or losec or nexium or prilosec or rapinex or zegerid or ocid or Lomac or Omepral or Omez).tw.
-
(pantoprazole or protium or protonix or Pantotab or Pantopan or Pantozol or Pantor or Pantoloc or Astropan or Controloc or Pantecta or Inipomp or Somac or Pantodac or Zurcal or Zentro).tw.
-
(lansoprazole or lanzoprazole or ag 1749 or agopton or bamalite or lanzor or monolitum or ogast or ogastro or opiren or prevacid or prezal or pro ulco or promeco or takepron or ulpax or zoton).tw.
-
(rabeprazole or aciphex or dexrabeprazole or e 3810 or ly‐307640 or pariet).tw.
-
(esomeprazole or nexium).tw.
-
(dexlansoprazole or Kapidex or Dexilant).tw.
-
(tenatoprazole or CAS 113712‐98‐4 or STU‐Na).tw. or exp tenatoprazole/
-
or/23‐31
-
exp cimetidine/ or exp histamine H2 receptor antagonist/ or exp famotidine/ or exp ranitidine/ or exp nizatidine/
-
(H2 receptor antagonist* or H2RA* or H2‐RA).tw.
-
(ranitidine or zantac or famotidine or pepcid or cimetidine or tagamet or nizatidine or Axid).tw.
-
histamine2 receptor antagonist.tw.
-
or/33‐36
-
exp bismuth citrate/ or exp bismuth salt/ or exp bismuth/ or exp bismuth salicylate/ or exp ranitidine bismuth citrate/ or exp colloidal bismuth compound/ or exp bismuth citrate plus metronidazole plus tetracycline/
-
exp amoxicillin plus clarithromycin plus lansoprazole/ or exp clarithromycin/ or exp clarithromycin derivative/
-
exp amoxicillin/
-
exp nitroimidazole/
-
exp macrolide/
-
(metronidazole or tinidazole or amoxicillin or amoxycillin).ti,ab,kw.
-
(clarithromycin or azithromycin or roxithromycin).ti,ab,kw.
-
(bismuth or nitroimidazole* or macrolide*).ti,ab,kw.
-
or/38‐45
-
32 or 37 or 46
-
22 and 47

Study flow diagram for RCTs

'Risk of bias' graph: review authors' judgements about each 'Risk of bias' item presented as percentages across all included studies.

'Risk of bias' summary: review authors' judgements about each 'Risk of bias' item for each included study.

Comparison 1 H. pylori eradication vs control ‐ main analyses, Outcome 1 Incidence of gastric cancer ‐ modified ITT analysis.
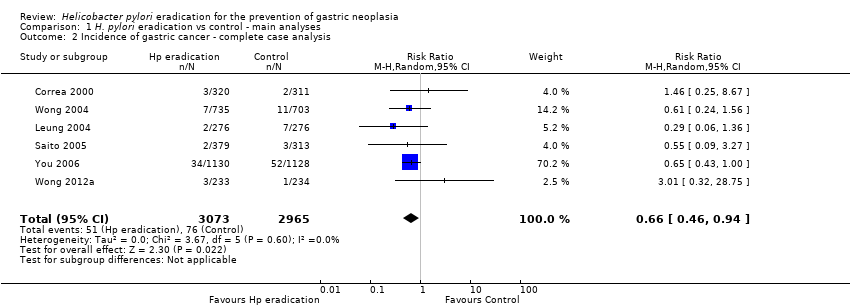
Comparison 1 H. pylori eradication vs control ‐ main analyses, Outcome 2 Incidence of gastric cancer ‐ complete case analysis.

Comparison 1 H. pylori eradication vs control ‐ main analyses, Outcome 3 Death from gastric cancer ‐ modified ITT analysis.

Comparison 1 H. pylori eradication vs control ‐ main analyses, Outcome 4 Death from all causes ‐ modified ITT analysis.

Comparison 1 H. pylori eradication vs control ‐ main analyses, Outcome 5 Incidence of oesophageal squamous cell carcinoma ‐ modified ITT analysis.

Comparison 2 H. pylori eradication vs control ‐ subgroup analysis according to presence or absence of pre‐neoplastic lesions at baseline, Outcome 1 Incidence of gastric cancer according to presence or absence of pre‐neoplastic lesions at baseline.

Comparison 3 H. pylori eradication vs control ‐ subgroup analysis according to use of vitamins or antioxidants, Outcome 1 Incidence of gastric cancer according to use of vitamins or anti‐oxidants.

Comparison 4 H. pylori eradication vs control ‐ sensitivity analyses, Outcome 1 Incidence of gastric cancer ‐ modified ITT analysis substituting the 10‐year follow‐up data from Zhou 2008 with the 5‐year follow‐up data from Leung 2004.
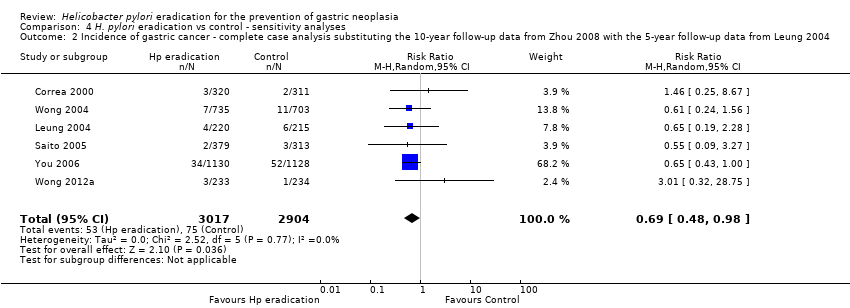
Comparison 4 H. pylori eradication vs control ‐ sensitivity analyses, Outcome 2 Incidence of gastric cancer ‐ complete case analysis substituting the 10‐year follow‐up data from Zhou 2008 with the 5‐year follow‐up data from Leung 2004.

Comparison 4 H. pylori eradication vs control ‐ sensitivity analyses, Outcome 3 Incidence of gastric cancer‐ modified ITT analysis including the two arms of celecoxib from Wong 2012.
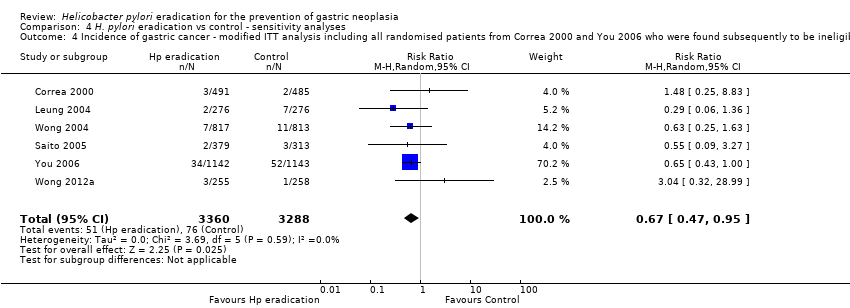
Comparison 4 H. pylori eradication vs control ‐ sensitivity analyses, Outcome 4 Incidence of gastric cancer ‐ modified ITT analysis including all randomised patients from Correa 2000 and You 2006 who were found subsequently to be ineligible or did not receive treatment.

Comparison 4 H. pylori eradication vs control ‐ sensitivity analyses, Outcome 5 Incidence of gastric cancer ‐ missing data imputation based on various assumptions.
| H. pylori eradication therapy compared to control for the prevention of gastric neoplasia in healthy asymptomatic infected individuals | ||||||
| Patient or population: healthy asymptomatic H. pylori‐infected individuals | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Control | H. pylori eradication therapy to prevent subsequent gastric cancer | |||||
| Incidence of gastric cancer ‐ modified ITT analysis | 24 per 1000 | 16 per 1000 | RR 0.66 | 6497 | ⊕⊕⊕⊝ | Number needed to treat to benefit was 124 (95% CI 78 to 843) |
| Death from gastric cancer ‐ modified ITT analysis | 16 per 1000 | 11 per 1000 | RR 0.67 | 4475 | ⊕⊕⊕⊝ | |
| Death from all causes ‐ modified ITT analysis | 67 per 1000 | 73 per 1000 | RR 1.09 | 5253 | ⊕⊕⊕⊝ | |
| Incidence of oesophageal squamous cell carcinoma ‐ modified ITT analysis | 1 per 1000 | 2 per 1000 | RR 1.99 | 1630 | ⊕⊕⊕⊝ | |
| Adverse events | See comment | See comment | Not estimable | 0 | See comment | Adverse events were poorly reported across the studies and could not be summarised. |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 As all but one study was conducted in East Asia, it is not possible to assess the effect of searching for and eradicating H. pylori in Western populations. | ||||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Incidence of gastric cancer ‐ modified ITT analysis Show forest plot | 6 | 6497 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.46, 0.95] |
| 2 Incidence of gastric cancer ‐ complete case analysis Show forest plot | 6 | 6038 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.46, 0.94] |
| 3 Death from gastric cancer ‐ modified ITT analysis Show forest plot | 3 | 4475 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.40, 1.11] |
| 4 Death from all causes ‐ modified ITT analysis Show forest plot | 4 | 5253 | Risk Ratio (M‐H, Random, 95% CI) | 1.09 [0.86, 1.38] |
| 5 Incidence of oesophageal squamous cell carcinoma ‐ modified ITT analysis Show forest plot | 1 | 1630 | Risk Ratio (M‐H, Random, 95% CI) | 1.99 [0.18, 21.91] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Incidence of gastric cancer according to presence or absence of pre‐neoplastic lesions at baseline Show forest plot | 6 | 6481 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.45, 1.17] |
| 1.1 Patients with precancerous lesions | 4 | 3425 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.47, 1.59] |
| 1.2 Patients with out precancerous lesions | 2 | 1812 | Risk Ratio (M‐H, Random, 95% CI) | 0.42 [0.02, 7.69] |
| 1.3 Mixed patients with and without precancerous lesions or ulcer (can't separate) | 2 | 1244 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.12, 1.23] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Incidence of gastric cancer according to use of vitamins or anti‐oxidants Show forest plot | 6 | 6497 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.45, 1.01] |
| 1.1 Without antioxidants | 6 | 4160 | Risk Ratio (M‐H, Random, 95% CI) | 0.82 [0.46, 1.45] |
| 1.2 With antioxidants | 2 | 2337 | Risk Ratio (M‐H, Random, 95% CI) | 0.52 [0.31, 0.87] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Incidence of gastric cancer ‐ modified ITT analysis substituting the 10‐year follow‐up data from Zhou 2008 with the 5‐year follow‐up data from Leung 2004 Show forest plot | 6 | 6532 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.49, 0.98] |
| 2 Incidence of gastric cancer ‐ complete case analysis substituting the 10‐year follow‐up data from Zhou 2008 with the 5‐year follow‐up data from Leung 2004 Show forest plot | 6 | 5921 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.48, 0.98] |
| 3 Incidence of gastric cancer‐ modified ITT analysis including the two arms of celecoxib from Wong 2012 Show forest plot | 6 | 7008 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.48, 0.97] |
| 4 Incidence of gastric cancer ‐ modified ITT analysis including all randomised patients from Correa 2000 and You 2006 who were found subsequently to be ineligible or did not receive treatment Show forest plot | 6 | 6648 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.47, 0.95] |
| 5 Incidence of gastric cancer ‐ missing data imputation based on various assumptions Show forest plot | 6 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 5.1 Assuming incidence of gastric cancer for missing participants in both arms same as observed in the trial control arm | 6 | 6497 | Risk Ratio (M‐H, Random, 95% CI) | 0.66 [0.47, 0.94] |
| 5.2 Assuming incidence of gastric cancer for missing participants in treatment arm same as observed in the trial control arm, but no new gastric cancer cases in the control arm among those with missing data | 6 | 6497 | Risk Ratio (M‐H, Random, 95% CI) | 0.69 [0.48, 0.98] |

