Contenido relacionado
Revisiones y protocolos relacionados
Roberto Ferrara, Martina Imbimbo, Reem Malouf, Sophie Paget-Bailly, François Calais, Corynne Marchal, Virginie Westeel | 30 abril 2021
Quimioterapia para el cáncer de pulmón de células no pequeñas avanzado en pacientes de edad avanzada
Fábio N Santos, Tiago B de Castria, Marcelo RS Cruz, Rachel Riera | 20 octubre 2015
Catherine Delbaldo, Stefan Michiels, Estelle Rolland, Nathalie Syz, Jean‐Charles Soria, Thierry Le Chevalier, Jean Pierre Pignon | 18 abril 2012
Non‐Small Cell Lung Cancer Collaborative Group | 12 mayo 2010
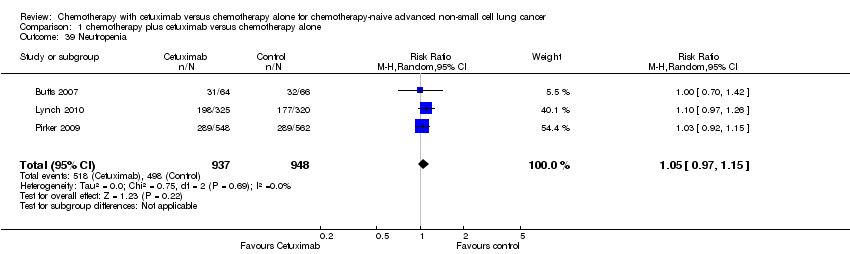
Sarah Burdett, Jean Pierre Pignon, Jayne Tierney, Helene Tribodet, Lesley Stewart, Cecile Le Pechoux, Anne Aupérin, Thierry Le Chevalier, Richard J Stephens, Rodrigo Arriagada, Julian PT Higgins, David H Johnson, Jan Van Meerbeeck, Mahesh KB Parmar, Robert L Souhami, Bengt Bergman, Jean‐Yves Douillard, Ariane Dunant, Chiaki Endo, David Girling, Harubumi Kato, Steven M Keller, Hideki Kimura, Aija Knuuttila, Ken Kodama, Ritsuko Komaki, Mark G Kris, Thomas Lad, Tommaso Mineo, Steven Piantadosi, Rafael Rosell, Giorgio Scagliotti, Lesley K Seymour, Frances A Shepherd, Richard Sylvester, Hirohito Tada, Fumihiro Tanaka, Valter Torri, David Waller, Ying Liang, for the Non‐Small Cell Lung Cancer Collaborative Group | 2 marzo 2015
Esther HA Sim, Ian A Yang, Richard Wood‐Baker, Rayleen V Bowman, Kwun M Fong | 16 enero 2018
Vitor F Vasconcellos, Guilherme N Marta, Edina MK da Silva, Aecio FT Gois, Tiago B de Castria, Rachel Riera | 13 enero 2020
Marta Pelayo Alvarez, Virginie Westeel, Marcela Cortés‐Jofré, Xavier Bonfill Cosp | 27 noviembre 2013
Janette Greenhalgh, Angela Boland, Victoria Bates, Fabio Vecchio, Yenal Dundar, Marty Chaplin, John A Green | 18 marzo 2021
for the Non‐Small Cell Lung Cancer Collaborative Group | 24 abril 2000
Respuestas clínicas Cochrane
Simone Mocellin | 19 noviembre 2015