Estimulación eléctrica neuromuscular para la debilidad muscular en pacientes adultos con enfermedades avanzadas
Información
- DOI:
- https://doi.org/10.1002/14651858.CD009419.pub3Copiar DOI
- Base de datos:
-
- Cochrane Database of Systematic Reviews
- Versión publicada:
-
- 17 octubre 2016see what's new
- Tipo:
-
- Intervention
- Etapa:
-
- Review
- Grupo Editorial Cochrane:
-
Grupo Cochrane de Dolor y cuidados paliativos
- Copyright:
-
- Copyright © 2016 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Cifras del artículo
Altmetric:
Citado por:
Autores
Contributions of authors
All authors were involved in the drafting of the protocol and final review. MM and AW developed the search strategy and searched for and obtained copies of studies for potential inclusion. SJ, WG, AW, and MM selected studies for inclusion, and all authors extracted data from studies and assessed risk of bias. SJ, MM, and WG entered data into Review Manager 5 (RevMan 2014), carried out analyses, and performed the GRADE assessments. All authors interpreted findings and approved the final review manuscript. MM is responsible for conducting any future updates.
Sources of support
Internal sources
-
Department of Palliative Medicine, Nottingham University Hospitals NHS Trust, UK.
AW is employed by Nottingham University Hospitals NHS Trust.
-
King's College London, Cicely Saunders Institute, Division of Palliative Care, Policy & Rehabilitation, UK.
MM, WG, and IJH are employed by King's College London.
-
Royal Brompton & Harefield NHS Foundation Trust and Imperial College, UK.
SJ and WD‐CM are employed by the Royal Brompton & Harefield NHS Foundation Trust.
External sources
-
National Institute of Health Research, UK.
IJH is a NIHR Senior Investigator. MM is supported by a NIHR post‐doctoral fellowship and a NIHR Clinical Trials Fellowship. MM and IJH are supported by the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) for South London. WD‐CM is supported by the NIHR Collaboration for Leadership in Applied Health Research and Care (CLAHRC) for North West London, NIHR Clinician Scientist Award, NIHR Clinical Trials Fellowship, and the NIHR Respiratory Biomedical Research Unit, Royal Brompton & Harefield NHS Foundation Trust and Imperial College, London, UK.
-
Cicely Saunders International, UK.
MM and IJH are supported by Cicely Saunders International.
-
Medical Research Council, UK.
WD‐CM is supported by a Medical Research Council (UK) New Investigator Research Grant.
Declarations of interest
SJ: none known. SJ is a physiotherapist and manages patients with respiratory conditions.
WD‐CM is a consultant chest physician and manages patients with respiratory conditions. He has received reimbursement for travel and accommodation costs from Boehringher Ingelheim arising from attendance at the European Respiratory Society Congress meeting in 2013.
WG: none known. WG coauthored one of the studies included in this review (Maddocks 2016a). She was not involved in the data extraction or 'Risk of bias' assessment for this study.
IJH: none known. IJH is a consultant palliative care physician and manages patients with advanced and/or progressive conditions.
AW: none known. AW is a consultant palliative care physician and manages patients with advanced and/or progressive conditions. He coauthored two of the studies included in this review (Maddocks 2009a; Maddocks 2013). He was not involved in the data extraction or 'Risk of bias' assessment for these studies.
MM: none known. MM coauthored three of the studies included in this review (Maddocks 2009a; Maddocks 2013; Maddocks 2016a). He was not involved in the data extraction or 'Risk of bias' assessment for these studies.
Acknowledgements
The 2016 update was undertaken with support from Anna Erskine, Managing Editor, Cochrane Pain Palliative and Supportive Care (PaPaS) Review Group. The search strategy was developed with the assistance of Joanne Abbott, Information Specialist, Cochrane PaPaS Group. Dr Hristina Petkova helped with the eligibility assessment for one excluded study published in Russian (Sumin 2009a).
Cochrane Review Group funding acknowledgement: The National Institute for Health Research (NIHR) is the largest single funder of the Cochrane PaPaS Group. Disclaimer: The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the NIHR, National Health Service (NHS), or the Department of Health.
Version history
| Published | Title | Stage | Authors | Version |
| 2016 Oct 17 | Neuromuscular electrical stimulation for muscle weakness in adults with advanced disease | Review | Sarah Jones, William D‐C Man, Wei Gao, Irene J Higginson, Andrew Wilcock, Matthew Maddocks | |
| 2013 Jan 31 | Neuromuscular electrical stimulation for muscle weakness in adults with advanced disease | Review | Matthew Maddocks, Wei Gao, Irene J Higginson, Andrew Wilcock | |
| 2011 Nov 09 | Neuromuscular electrical stimulation for muscle weakness in adults with advanced disease | Protocol | Matthew Maddocks, Wei Gao, Irene J Higginson, Andrew Wilcock | |
Differences between protocol and review
In this 2016 updated review we considered study size as a new 'Risk of bias' item to check for possible bias from small study size. We also included GRADE assessments of the quality of the evidence and added a 'Summary of findings' table. We did not include studies examining the acute effects of NMES following a single session.
Notes
A new search within two years is not likely to identify any potentially relevant studies likely to change the conclusions. Therefore, this review has now been stabilised following discussion with the authors and editors. The review will be re‐assessed for updating in five years. If appropriate, we will update the review before this date if new evidence likely to change the conclusions is published, or if standards change substantially which necessitate major revisions.
Keywords
MeSH
Medical Subject Headings (MeSH) Keywords
- Chronic Disease;
- Disease Progression;
- Heart Failure [complications];
- Leg;
- Muscle Strength;
- Muscle Weakness [etiology, *therapy];
- Muscle, Skeletal [anatomy & histology];
- Physical Exertion [physiology];
- Pulmonary Disease, Chronic Obstructive [complications];
- Quadriceps Muscle [physiology];
- Randomized Controlled Trials as Topic;
- Respiration Disorders [complications];
- Thoracic Neoplasms [complications];
- Transcutaneous Electric Nerve Stimulation [adverse effects, *methods];
Medical Subject Headings Check Words
Adult; Humans;
PICO

Study flow diagram.

'Risk of bias' graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
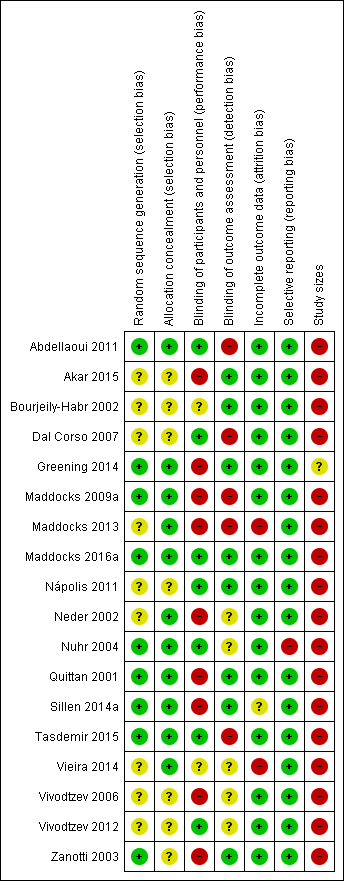
'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.

Forest plot of quadriceps muscle strength for NMES versus control.
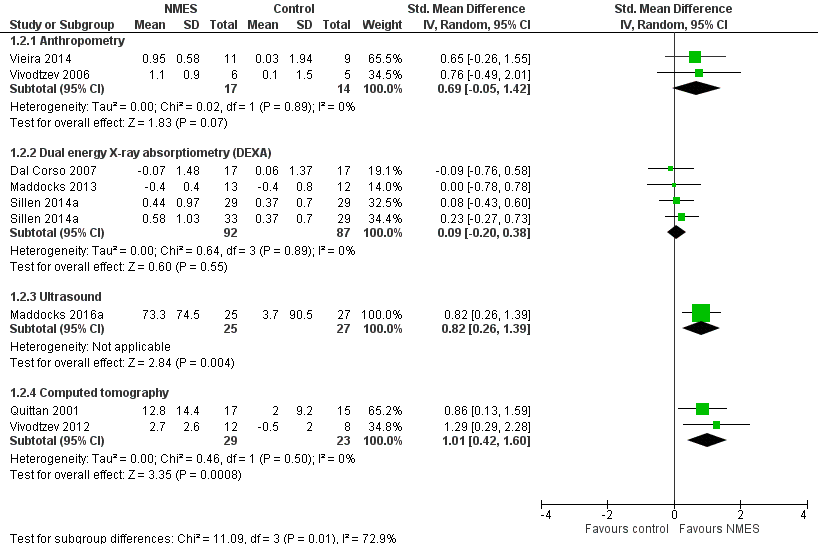
Forest plot of muscle mass for NMES versus control.

Forest plot of exercise performance for NMES versus control.

Comparison 1 Neuromuscular electrical stimulation versus control, Outcome 1 Quadriceps muscle strength.

Comparison 1 Neuromuscular electrical stimulation versus control, Outcome 2 Muscle mass.

Comparison 1 Neuromuscular electrical stimulation versus control, Outcome 3 Exercise performance.
| NMES for adults with advanced disease for muscle weakness | ||||
| Patient or population: adults with advanced disease for muscle weakness Control: no intervention (7 studies), placebo NMES (8 studies), or resistance training (1 study) | ||||
| Outcomes | Illustrative comparative risks* (95% CI) | No of Participants | Quality of the evidence | |
| Assumed risk | Corresponding risk | |||
| Control | NMES | |||
| Quadriceps muscle strength | The mean change was 0.43 standard deviations from baseline. | The mean change in the intervention groups was 0.53 standard deviations higher (ranging from 0.19 to 0.87 standard deviations higher). | 781 | ⊕⊕⊝⊝ |
| Safety | No serious adverse events related to control interventions reported. | No serious adverse events related to NMES reported. | 933 | ⊕⊕⊕⊝ |
| Safety Adverse events: Muscle discomfort | 0/415 (0%) participants reported muscle discomfort following control interventions. | 19/518 (3.7%) participants reported muscle discomfort following NMES. | 933 (18 studies) | ⊕⊕⊕⊝ |
| Muscle mass | The mean change in muscle mass ranged from 0.04 to 0.49 standard deviations from baseline across the different assessment modalities used. | The mean change in muscle mass ranged from 0.09 to 1.01 standard deviations higher across the different assessment modalities used. | 314 | ⊕⊝⊝⊝ |
| Exercise performance ‐ walking distance | The mean change in distance walked was 21, 36, and 37 metres from baseline across the different walking tests used. | The mean change in distance walked was 35, 9, and 64 metres further across the different walking tests used. | 788 | ⊕⊝⊝⊝ |
| Exercise performance ‐ peak oxygen uptake | The mean change in peak oxygen uptake was ‐0.4 mL/min from baseline. | The mean exercise performance ‐ peak oxygen uptake in the intervention groups was 44.8 mL/min higher (95% CI 7.3 lower to 97.0 higher) | 109 | ⊕⊕⊝⊝ |
| *The basis for the assumed risk is the mean change from baseline in the control groups. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||
| GRADE Working Group grades of evidence | ||||
| 1Downgraded once: the lower 95% CI for the estimate of effect was below what would be considered a small effect (standardised mean difference 0.2). | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Quadriceps muscle strength Show forest plot | 12 | 781 | Std. Mean Difference (IV, Random, 95% CI) | 0.53 [0.19, 0.87] |
| 2 Muscle mass Show forest plot | 8 | Std. Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 2.1 Anthropometry | 2 | 31 | Std. Mean Difference (IV, Random, 95% CI) | 0.69 [‐0.05, 1.42] |
| 2.2 Dual energy X‐ray absorptiometry (DEXA) | 3 | 179 | Std. Mean Difference (IV, Random, 95% CI) | 0.09 [‐0.20, 0.38] |
| 2.3 Ultrasound | 1 | 52 | Std. Mean Difference (IV, Random, 95% CI) | 0.82 [0.26, 1.39] |
| 2.4 Computed tomography | 2 | 52 | Std. Mean Difference (IV, Random, 95% CI) | 1.01 [0.42, 1.60] |
| 3 Exercise performance Show forest plot | 13 | Mean Difference (IV, Random, 95% CI) | Subtotals only | |
| 3.1 6‐minute walk test (m) (6MWT) | 7 | 317 | Mean Difference (IV, Random, 95% CI) | 34.78 [13.52, 56.05] |
| 3.2 Incremental shuttle walk test (m) (ISWT) | 3 | 434 | Mean Difference (IV, Random, 95% CI) | 8.72 [‐34.87, 52.31] |
| 3.3 Endurance shuttle walk test (m) (ESWT) | 4 | 452 | Mean Difference (IV, Random, 95% CI) | 64.13 [‐17.79, 146.05] |
| 3.4 Cardiopulmonary exercise testing (mL/min) (CPET) | 4 | 109 | Mean Difference (IV, Random, 95% CI) | 44.82 [‐7.34, 96.99] |

