Inmunoglobulina anti‐hepatitis B durante el embarazo para la prevención de la transmisión maternoinfantil del virus de la hepatitis B
Appendices
Appendix 1. Search strategies
| Database | Time span | Search strategies |
| The Cochrane Hepato‐Biliary Group Controlled Trials Register | June 2016. | (hepatitis B OR hep b OR HBV OR immune globulin OR HBIG) AND (pregnan* OR mother OR maternal OR child OR baby OR perinatal) AND transmission |
| Cochrane Central Register of Controlled Trials (CENTRAL) in the Cochrane Library | 2016, Issue 5 | #1 MeSH descriptor: [Hepatitis B] explode all trees #2 hepatitis B or hep b or HBV or immune globulin or HBIG #3 #1 or #2 #4 MeSH descriptor: [Pregnancy] explode all trees #5 MeSH descriptor: [Prenatal Diagnosis] explode all trees #6 pregnan* #7 #4 or #5 or #6 #8 MeSH descriptor: [Infectious Disease Transmission, Vertical] explode all trees #9 (mother or maternal or child or baby or perinatal) and transmission #10 #8 or #9 #11 #3 and #7 and #10 |
| MEDLINE Ovid | 1946 to June 2016. | 1. exp Hepatitis B/ 2. (hepatitis B or hep b or HBV or immune globulin or HBIG).mp. [mp=title, original title, abstract, name of substance word, subject heading word, unique identifier] 3. 1 or 2 4. exp Pregnancy/ 5. exp Prenatal Diagnosis/ 6. pregnan*.mp. [mp=title, original title, abstract, name of substance word, subject heading word, unique identifier] 7. 4 or 5 or 6 8. exp Infectious Disease Transmission, Vertical/ 9. ((mother or maternal or child or baby or perinatal) and transmission).mp. [mp=title, original title, abstract, name of substance word, subject heading word, unique identifier] 10. 8 or 9 11. 3 and 7 and 10 12. (random* or blind* or placebo* or meta‐analysis).mp. [mp=title, original title, abstract, name of substance word, subject heading word, unique identifier] 13. 11 and 12 |
| Embase Ovid | 1974 to June 2016. | 1. exp hepatitis B/ 2. (hepatitis B or hep b or HBV or immune globulin or HBIG).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] 3. 1 or 2 4. exp pregnancy/ 5. exp prenatal diagnosis/ 6. pregnan*.mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] 7. 4 or 5 or 6 8. exp vertical transmission/ 9. ((mother or maternal or child or baby or perinatal) and transmission).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] 10. 8 or 9 11. 3 and 7 and 10 12. (random* or blind* or placebo* or meta‐analysis).mp. [mp=title, abstract, subject headings, heading word, drug trade name, original title, device manufacturer, drug manufacturer name] 13. 11 and 12 |
| Science Citation Index Expanded (Web of Science) | 1900 to June 2016. | # 6 #5 AND #4 |
| SCOPUS | 1966 to June 2016. | # 6 #5 AND #4 |
| African Journals OnLine | 1998 to June 2016. | # 6 #5 AND #4 |
| INDEX MEDICUS | 1879 to June 2016. | # 6 #5 AND #4 |
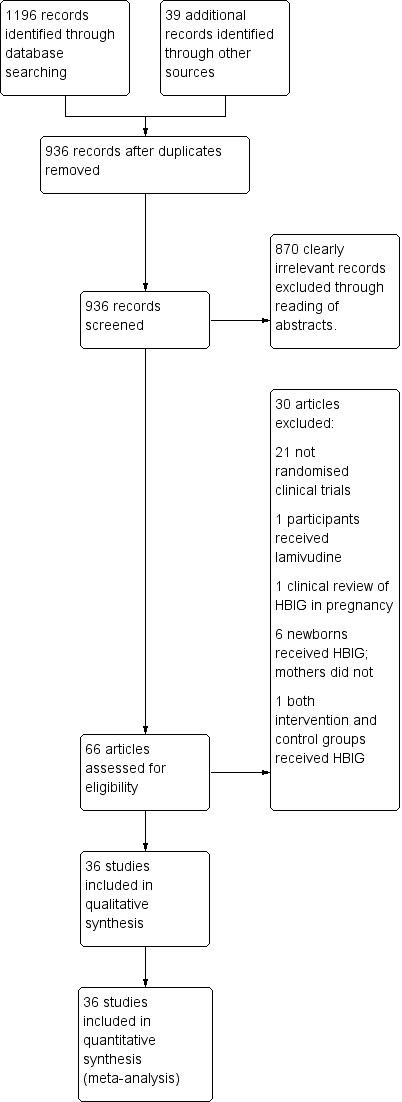
Study flow diagram for searches on hepatitis B Immunoglobulin (HBIG).

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
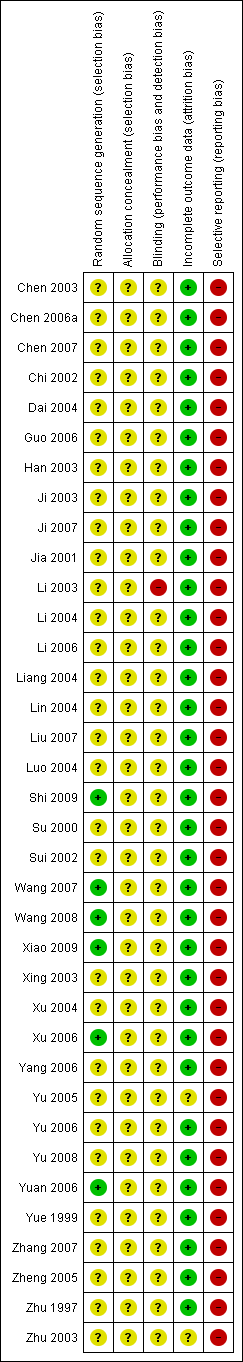
Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
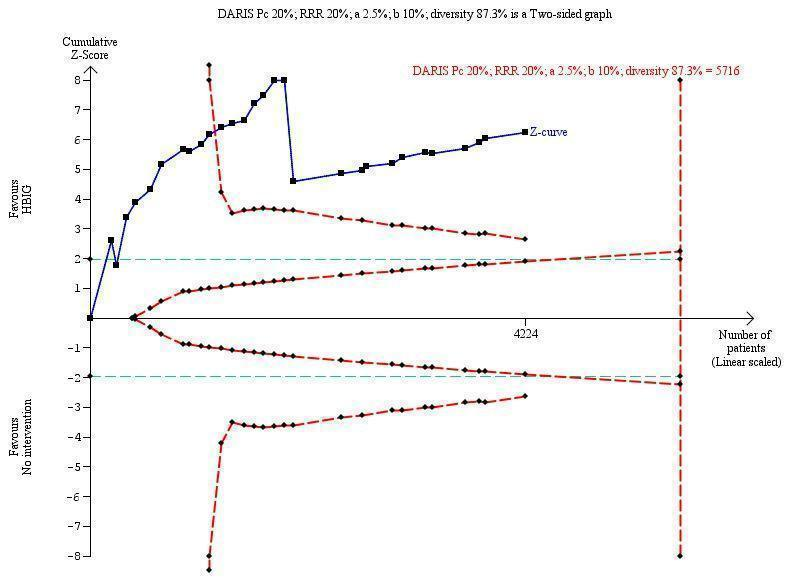
Trial Sequential Analysis (TSA) of the random‐effects meta‐analysis of the effect of hepatitis B immunoglobulin (HBIG) versus no intervention on the number of newborns with HBsAg‐positive results at end of follow‐up. The diversity‐adjusted required information size (DARIS) of 5716 participants was calculated based upon a proportion of 20% of babies tested positive for HBsAg in the control group, a relative risk reduction of a 20% in HBIG group, an alpha (type I error) of 2.5%, a beta (type II error) of 10%, and a diversity (D) of 87.3%. The actually accrued number of participants is 4224, which is 74% of the DARIS. The solid blue curve presents the cumulative meta‐analysis Z‐score and the inward sloping red curves present the adjusted threshold for statistical significance according to the two‐sided Lan‐DeMets trial sequential monitoring boundaries. The blue cumulative Z‐curve crosses the red trial sequential monitoring boundary for benefit during the 11th trial. This implies that there is no risk of random error in the estimate of a beneficial effect of HBIG versus no intervention on the number of newborns with HBsAg‐positive results at end of follow‐up. The TSA‐adjusted confidence interval is 0.20 to 0.52.
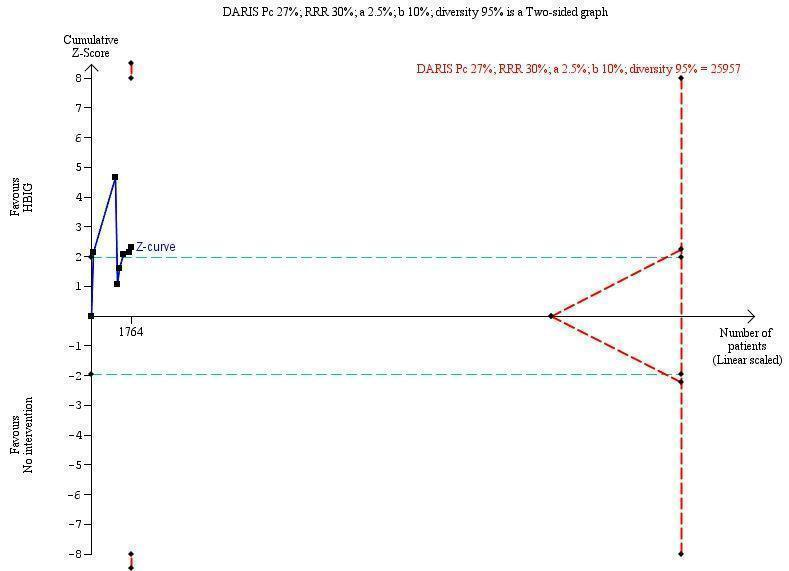
Trial Sequential Analysis (TSA) of the random‐effects meta‐analysis of the effect of hepatitis B immunoglobulin (HBIG) versus no intervention on the number of newborns with hepatitis B envelope antigen (HBeAg)‐positive results at end of follow‐up. The diversity‐adjusted required information size (DARIS) of 25,957 participants was calculated based upon a proportion of 27% of babies tested positive for HBeAg in the control group, a relative risk reduction of a 30% in HBIG group, an alpha (type I error) of 2.5%, a beta (type II error) of 10%, and a diversity (D) of 95%. The actually accrued number of participants is 1764, which is only 6.8% of the DARIS. (We planned to use a relative risk reduction of 20%, but this led to a DARIS of 60,715 participants and the TSA figure could not be drawn by the program; therefore, a relative risk reduction of 30% was adopted instead.) The solid blue curve presents the cumulative meta‐analysis Z‐score and the inward sloping red curves present the adjusted threshold for statistical significance according to the two‐sided Lan‐DeMets trial sequential monitoring boundaries. The blue cumulative Z‐curve does not cross the red inward sloping trial sequential monitoring boundaries for benefit or harm. Therefore, there is no evidence to support that HBIG influences number of newborns with HBeAg‐positive results at end of follow‐up. The cumulative Z‐curve does not reach the futility area, demonstrating that further trials are needed. The TSA‐adjusted confidence interval is wider than 0.04 to 6.37.
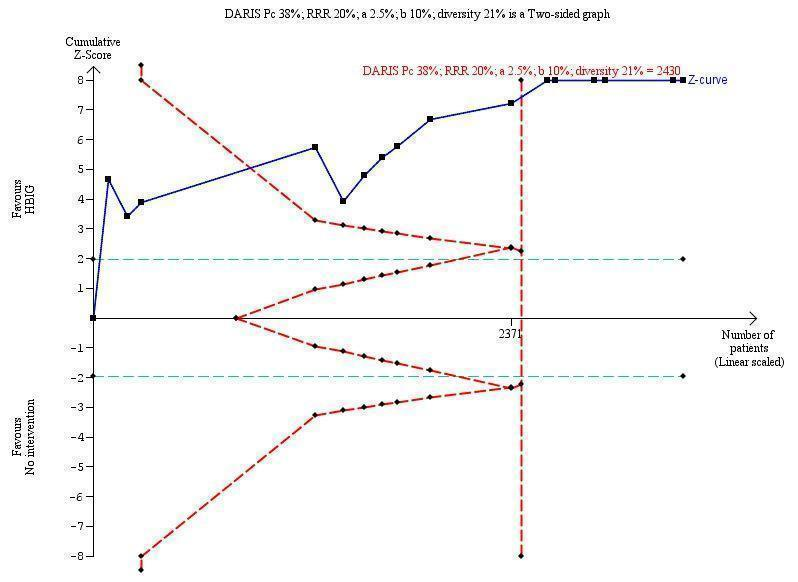
Trial Sequential Analysis (TSA) of the random‐effects meta‐analysis of the effect of hepatitis B immunoglobulin (HBIG) versus no intervention on the number of newborns with hepatitis B virus DNA (HBV‐DNA) positive results at end of treatment. The diversity‐adjusted required information size (DARIS) of n = 2430 participants was calculated based upon a proportion of 38% of babies tested positive for HBV‐DNA, a relative risk reduction of a 20% in HBIG group, an alpha (type I error) of 2.5%, a beta (type II error) of 10%, and a diversity (D) of 21%. The actually accrued number of participants is 2994, which is more than the DARIS of 2430 participants. The solid blue curve presents the cumulative meta‐analysis Z‐score and the inward sloping red curves present the adjusted threshold for statistical significance according to the two‐sided Lan‐DeMets trial sequential monitoring boundaries. The blue cumulative Z‐curve crosses the red trial sequential monitoring boundary for benefit during the fourth trial. This implies that there is no risk of random error in the estimate of a beneficial effect of HBIG versus no intervention on the number of newborns with HBV‐DNA positive results at end of treatment. The TSA‐adjusted and 95% confidence intervals is from 0.22 to 0.37.
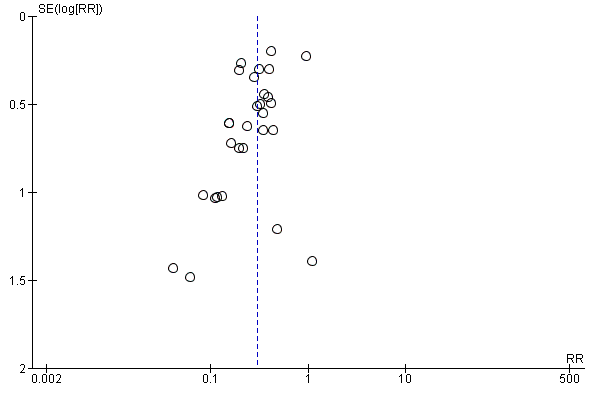
Funnel plot of comparison: 1 Hepatitis B immunoglobulin (HBIG) versus no intervention, outcome: 1.1 Newborn positive for HBsAg.
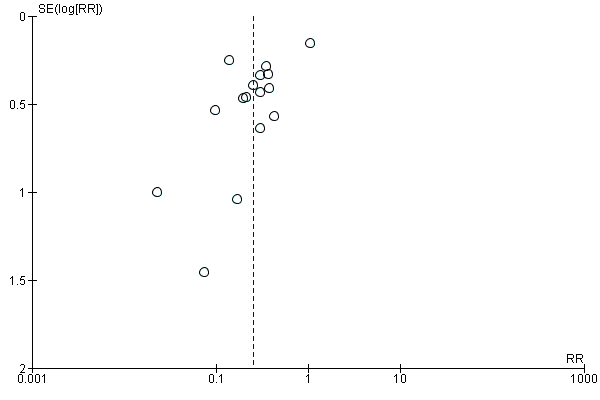
Funnel plot of comparison: 1 Hepatitis B immunoglobulin (HBIG) versus no intervention, outcome: 1.3 Newborn positive for HBV‐DNA.

Comparison 1 Hepatitis B immunoglobulin (HBIG) versus no intervention, Outcome 1 Newborn positive for HBsAg.
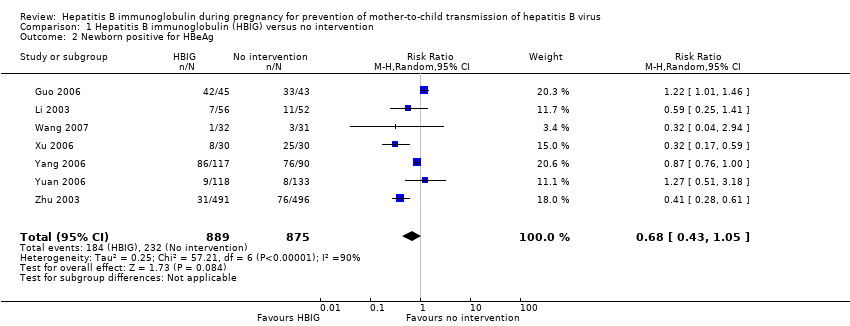
Comparison 1 Hepatitis B immunoglobulin (HBIG) versus no intervention, Outcome 2 Newborn positive for HBeAg.

Comparison 1 Hepatitis B immunoglobulin (HBIG) versus no intervention, Outcome 3 Newborn positive for HBV‐DNA.

Comparison 2 Hepatitis B immunoglobulin (HBIG) versus no intervention according to dosing regimen of HBIG administration, Outcome 1 Newborn positive for HBsAg.
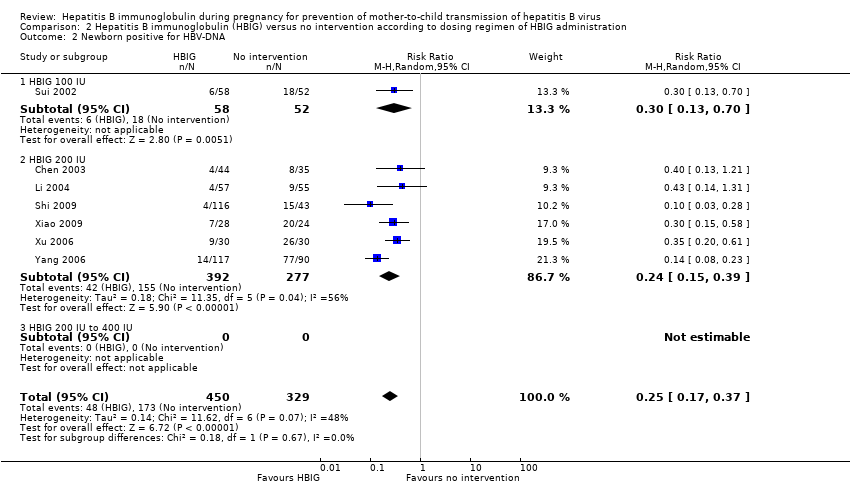
Comparison 2 Hepatitis B immunoglobulin (HBIG) versus no intervention according to dosing regimen of HBIG administration, Outcome 2 Newborn positive for HBV‐DNA.
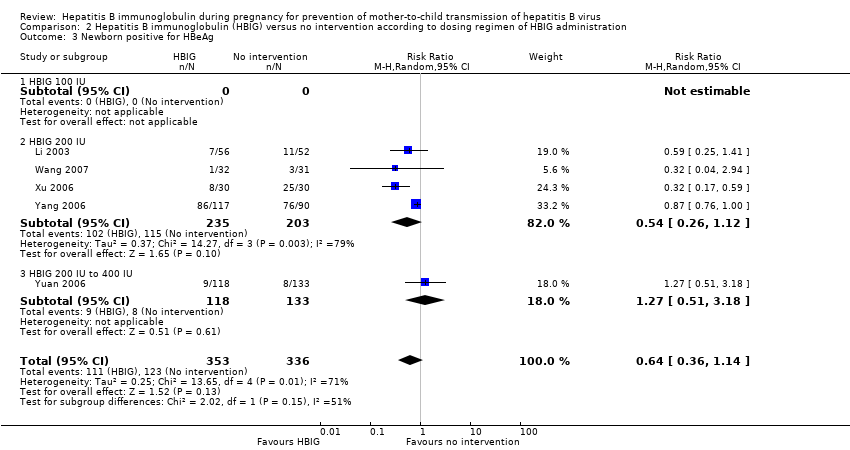
Comparison 2 Hepatitis B immunoglobulin (HBIG) versus no intervention according to dosing regimen of HBIG administration, Outcome 3 Newborn positive for HBeAg.
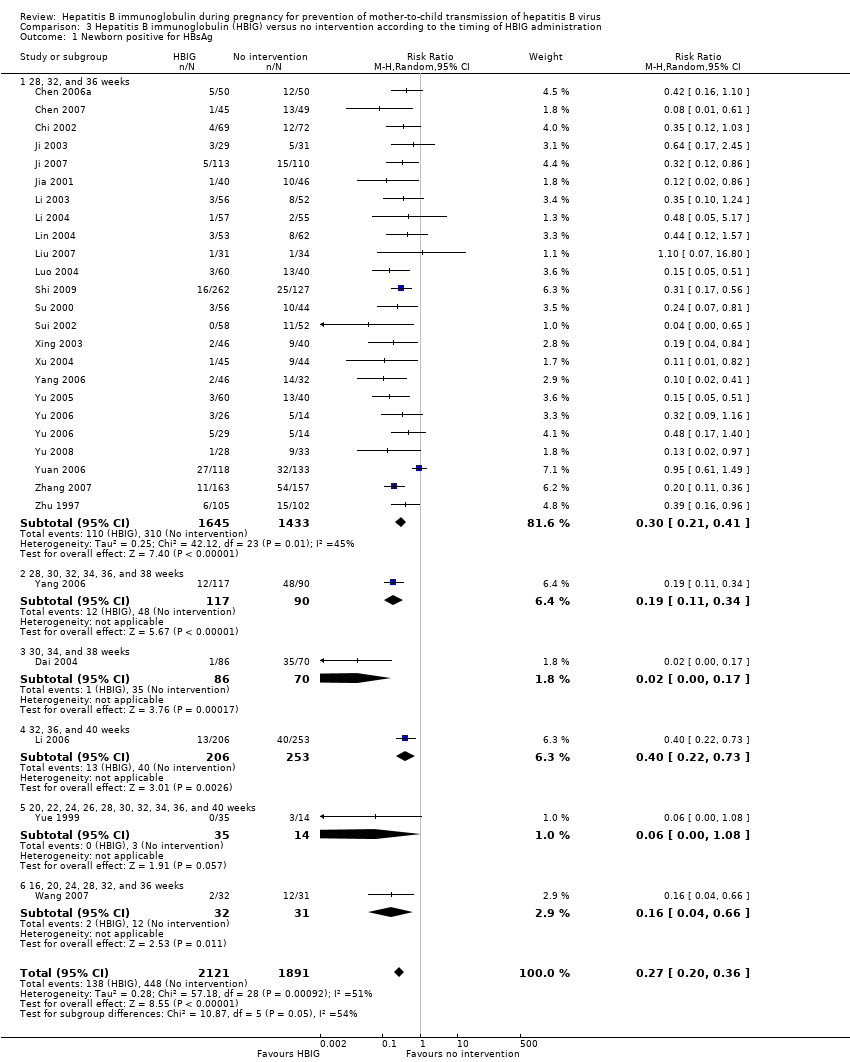
Comparison 3 Hepatitis B immunoglobulin (HBIG) versus no intervention according to the timing of HBIG administration, Outcome 1 Newborn positive for HBsAg.
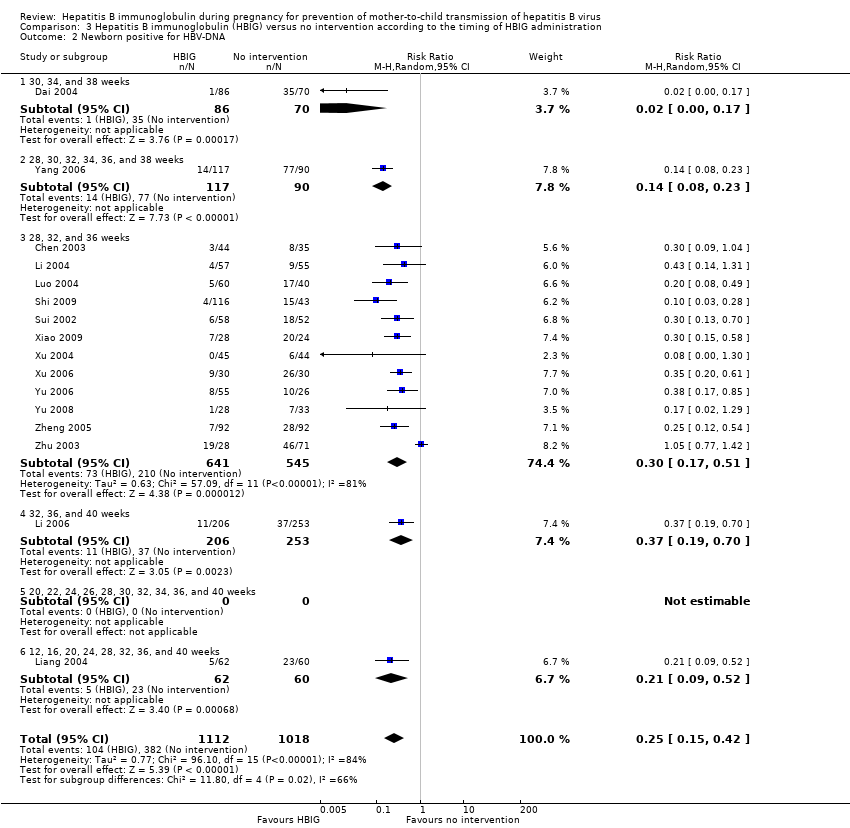
Comparison 3 Hepatitis B immunoglobulin (HBIG) versus no intervention according to the timing of HBIG administration, Outcome 2 Newborn positive for HBV‐DNA.
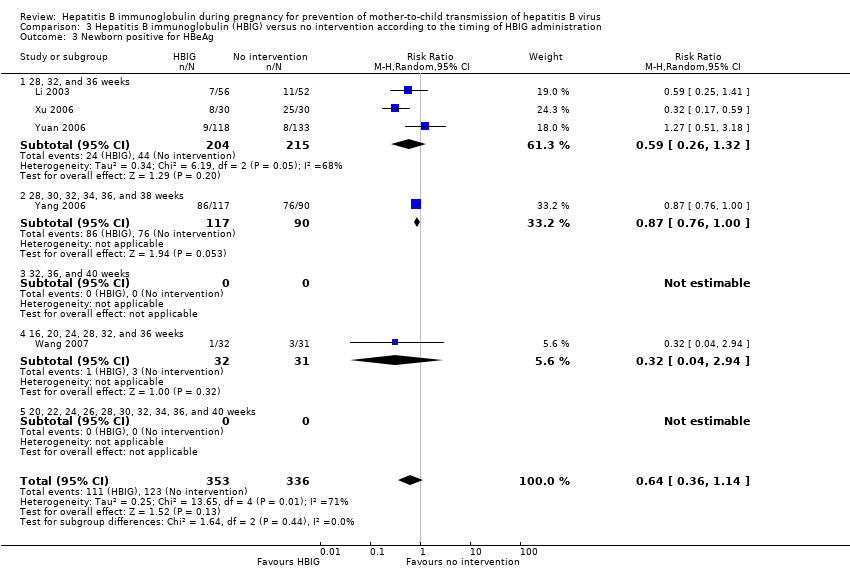
Comparison 3 Hepatitis B immunoglobulin (HBIG) versus no intervention according to the timing of HBIG administration, Outcome 3 Newborn positive for HBeAg.
| Hepatitis B immunoglobulin (HBIG) vs no intervention for prevention of mother‐to‐child transmission of hepatitis B virus | ||||||
| Participants: pregnant women positive for HBsAg or positive for HBeAg, or both. Comparison: no intervention. | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Control | HBIG versus no intervention | |||||
| All‐cause mortality or other serous adverse events of the newborn | Study population | Not estimable | 0 | See comment | ||
| See comment | See comment | |||||
| Moderate | ||||||
| All‐cause mortality or other serous adverse events of the mothers | Study population | Not estimable | 0 | See comment | ||
| See comment | See comment | |||||
| Moderate | ||||||
| Newborn with HBsAg‐positive result | Study population | RR 0.3 | 5310 | ⊕⊝⊝⊝ | ||
| 211 per 1000 | 63 per 1000 | |||||
| Moderate | ||||||
| 213 per 1000 | 64 per 1000 | |||||
| Newborn with HBeAg‐positive result | Study population | RR 0.68 | 1764 | ⊕⊝⊝⊝ | ||
| 265 per 1000 | 180 per 1000 | |||||
| Moderate | ||||||
| 212 per 1000 | 144 per 1000 | |||||
| Newborn with HBV‐DNA‐positive result | Study population | RR 0.25 | 2130 | ⊕⊕⊝⊝ | ||
| 375 per 1000 | 94 per 1000 | |||||
| Moderate | ||||||
| 366 per 1000 | 91 per 1000 | |||||
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Comment: Data for this outcome was not reported in any of the included trials. | ||||||
| Study ID | Study location | Participants | Interventions | Outcomes | Funding |
| Zhejiang | 79 participants (44 intervention, 35 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA | Not stated. | |
| Guangdong | 100 participants (50 intervention, 50 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Shenzhen | 94 participants (45 intervention, 49 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, anti‐HBs | Not stated. | |
| Zhejiang | 141 participants (69 intervention, 72 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Zhejiang | 156 participants (86 intervention, 70 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA, anti‐HBs | Not stated. | |
| Henan | 88 participants (45 intervention, 43 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, HBeAg anti‐HBs. | Not stated | |
| Guangdong | 216 participants (126 intervention, 90 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg. | Not stated. | |
| Zhejiang | 60 participants (29 intervention, 31 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, anti‐HBs, adverse events. | Not stated | |
| Shanghai | 223 participants (113 intervention, 110 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Jiangsu | 86 participants (40 intervention, 46 control) aged 22 to 32 years. | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Guangdong | 108 participants (56 intervention, 52 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg or HBeAg, or both. | Not stated. | |
| Guangdong | 112 participants (57 intervention, 55 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg or HBV‐DNA, or both. | Not stated. | |
| Hubei | 448 participants (202 intervention, 246 control) aged 18 to 38 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA, HBsAg. | Not stated. | |
| Guangdong | 122 participants (62 intervention, 60 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA. | Not stated. | |
| Shanghai | 117 participants (55 intervention, 62 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg. | Not stated. | |
| Henan | 86 participants (43 intervention, 43 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg. | Not stated. | |
| Jiangxi | 100 participants (60 intervention, 40 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA, HBsAg. | Not stated. | |
| Guangzhou | 389 participants (262 intervention, 127 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg or HBV‐DNA, or both | Research supported by GlaxoSmithKline Research and Development Grant NUC30914; Science and Research Foundations of Sun Yat‐Sen University and Guangzhou Science Committee, No 1999‐J‐005‐01. | |
| Henan | 98 participants (55 intervention, 43 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Shandong | 108 participants (56 intervention, 52 control) | Intervention: HBIG 100 IU. Control: no intervention. | Newborn HBsAg, HBV‐DNA, and anti‐HBs. | Not stated. | |
| Shandong | 63 participants (32 intervention, 31 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg and HBeAg | Not stated. | |
| Taizhou | 279 participants (159 intervention, 120 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Xinjiang | 52 participants (28 intervention, 24 control) | Intervention: HBIG 200 IU. Control: no intervention. | newborn HBV‐DNA. | Not stated. | |
| Henan | 86 participants (46 intervention, 40 control) aged 22 to 28 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Supported by Technology Research Fund Committee of Henan province (No. 981170112). | |
| Shandong | 88 participants (44 intervention, 44 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, HBV‐DNA and anti‐HBs. 8‐month‐old babies positive for HBsAg, HBV‐DNA, and anti‐HBs. Maximum duration of surveillance: 8 months. Follow‐up time point: 8 months after birth. | Not stated. | |
| Xinjiang | 52 participants (28 intervention, 24 control) | Intervention: HBIG 200 IU. Control: no intervention. | newborn HBeAg and HBV‐DNA. | Not stated. | |
| Jiangsu | 285 participants (163 intervention, 162 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, HBeAg and HBV‐DNA | Not stated. | |
| Guangdong | 100 participants (60 intervention, 40 control) | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBeAg, anti‐HBs | Not stated. | |
| Shanghai | 83 participants (26 intervention I, 29 intervention II, 28 control) aged 20 to 33 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA | Not stated. | |
| Guangxi | 61 participants (28 intervention, 33 control) aged 22 to 39 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA, HBsAg | Not stated. | |
| Huizhou | 250 participants (117 intervention, 113 control) | Intervention: HBIG 400 IU. Control: no intervention. | Newborn HBsAg, HBeAg, antibodies to HBsAg, HBeAg, and HBcAg; adverse effects of the immunoglobulins to the neonates and mothers | Supported by Huizhou Municipal Central hospital and Huizhou Science and Technology Bureau. | |
| Shanxi | 48 participants (34 intervention, 14 control) aged 20 to 33 years | Intervention: HBIG 100 IU. Control: no intervention. | Newborn HBsAg, anti‐HBs | Not stated. | |
| Guangdong | 320 participants (163 intervention, 157 control) aged 19 to 36 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg | Not stated. | |
| Guangdong | 184 participants (92 intervention, 92 control) aged 22 to 39 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBV‐DNA | Not stated. | |
| Shanghai | 204 participants (103 intervention, 101 control) aged 20 to 34 years | Intervention: HBIG 200 IU. Control: no intervention. | Newborn HBsAg, HBeAg, antibodies to HBsAg, HBeAg, and HBcAg. | Not stated. | |
| Shanghai | 980 participants (487 intervention, 493 control) aged 19 to 35 years | Intervention: HBIG 200 IU or 400 IU. Control: no intervention. | Newborn HBsAg, HBeAg, and HBV‐DNA. | Supported by a grant from the Ministry of Public Health China (No. 97030223). | |
| anti‐HBc: anti‐hepatitis core; anti‐HBe: anti‐hepatitis B envelope; anti‐HBs: anti‐hepatitis B surface; HBIG: hepatitis B immunoglobulin; HBcAg: hepatitis B core antigen; HBeAg: hepatitis B envelope antigen; HBsAg: hepatitis B surface antigen; HBV‐DNA: hepatitis B virus DNA. | |||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Newborn positive for HBsAg Show forest plot | 29 | 5310 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.24, 0.38] |
| 2 Newborn positive for HBeAg Show forest plot | 7 | 1764 | Risk Ratio (M‐H, Random, 95% CI) | 0.68 [0.43, 1.05] |
| 3 Newborn positive for HBV‐DNA Show forest plot | 16 | 2130 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.15, 0.42] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Newborn positive for HBsAg Show forest plot | 28 | 4281 | Risk Ratio (M‐H, Random, 95% CI) | 0.28 [0.21, 0.37] |
| 1.1 HBIG 100 IU | 2 | 159 | Risk Ratio (M‐H, Random, 95% CI) | 0.05 [0.01, 0.36] |
| 1.2 HBIG 200 IU | 25 | 3855 | Risk Ratio (M‐H, Random, 95% CI) | 0.26 [0.21, 0.33] |
| 1.3 HBIG 400 IU | 2 | 267 | Risk Ratio (M‐H, Random, 95% CI) | 0.67 [0.30, 1.53] |
| 2 Newborn positive for HBV‐DNA Show forest plot | 7 | 779 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.17, 0.37] |
| 2.1 HBIG 100 IU | 1 | 110 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.13, 0.70] |
| 2.2 HBIG 200 IU | 6 | 669 | Risk Ratio (M‐H, Random, 95% CI) | 0.24 [0.15, 0.39] |
| 2.3 HBIG 200 IU to 400 IU | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Newborn positive for HBeAg Show forest plot | 5 | 689 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.36, 1.14] |
| 3.1 HBIG 100 IU | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.2 HBIG 200 IU | 4 | 438 | Risk Ratio (M‐H, Random, 95% CI) | 0.54 [0.26, 1.12] |
| 3.3 HBIG 200 IU to 400 IU | 1 | 251 | Risk Ratio (M‐H, Random, 95% CI) | 1.27 [0.51, 3.18] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Newborn positive for HBsAg Show forest plot | 27 | 4012 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.20, 0.36] |
| 1.1 28, 32, and 36 weeks | 23 | 3078 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.21, 0.41] |
| 1.2 28, 30, 32, 34, 36, and 38 weeks | 1 | 207 | Risk Ratio (M‐H, Random, 95% CI) | 0.19 [0.11, 0.34] |
| 1.3 30, 34, and 38 weeks | 1 | 156 | Risk Ratio (M‐H, Random, 95% CI) | 0.02 [0.00, 0.17] |
| 1.4 32, 36, and 40 weeks | 1 | 459 | Risk Ratio (M‐H, Random, 95% CI) | 0.40 [0.22, 0.73] |
| 1.5 20, 22, 24, 26, 28, 30, 32, 34, 36, and 40 weeks | 1 | 49 | Risk Ratio (M‐H, Random, 95% CI) | 0.06 [0.00, 1.08] |
| 1.6 16, 20, 24, 28, 32, and 36 weeks | 1 | 63 | Risk Ratio (M‐H, Random, 95% CI) | 0.16 [0.04, 0.66] |
| 2 Newborn positive for HBV‐DNA Show forest plot | 16 | 2130 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.15, 0.42] |
| 2.1 30, 34, and 38 weeks | 1 | 156 | Risk Ratio (M‐H, Random, 95% CI) | 0.02 [0.00, 0.17] |
| 2.2 28, 30, 32, 34, 36, and 38 weeks | 1 | 207 | Risk Ratio (M‐H, Random, 95% CI) | 0.14 [0.08, 0.23] |
| 2.3 28, 32, and 36 weeks | 12 | 1186 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.17, 0.51] |
| 2.4 32, 36, and 40 weeks | 1 | 459 | Risk Ratio (M‐H, Random, 95% CI) | 0.37 [0.19, 0.70] |
| 2.5 20, 22, 24, 26, 28, 30, 32, 34, 36, and 40 weeks | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 2.6 12, 16, 20, 24, 28, 32, 36, and 40 weeks | 1 | 122 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.09, 0.52] |
| 3 Newborn positive for HBeAg Show forest plot | 5 | 689 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.36, 1.14] |
| 3.1 28, 32, and 36 weeks | 3 | 419 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.26, 1.32] |
| 3.2 28, 30, 32, 34, 36, and 38 weeks | 1 | 207 | Risk Ratio (M‐H, Random, 95% CI) | 0.87 [0.76, 1.00] |
| 3.3 32, 36, and 40 weeks | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |
| 3.4 16, 20, 24, 28, 32, and 36 weeks | 1 | 63 | Risk Ratio (M‐H, Random, 95% CI) | 0.32 [0.04, 2.94] |
| 3.5 20, 22, 24, 26, 28, 30, 32, 34, 36, and 40 weeks | 0 | 0 | Risk Ratio (M‐H, Random, 95% CI) | 0.0 [0.0, 0.0] |

