Aromaterapia para el tratamiento de las náuseas y los vómitos posoperatorios
Información
- DOI:
- https://doi.org/10.1002/14651858.CD007598.pub2Copiar DOI
- Base de datos:
-
- Cochrane Database of Systematic Reviews
- Versión publicada:
-
- 18 abril 2012see what's new
- Tipo:
-
- Intervention
- Etapa:
-
- Review
- Grupo Editorial Cochrane:
-
Grupo Cochrane de Anestesia
- Copyright:
-
- Copyright © 2012 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Cifras del artículo
Altmetric:
Citado por:
Autores
Contributions of authors
Conceiving the review: Sonia Hines (SH)
Designing the review: SH
Co‐ordinating the review: SH
Undertaking manual searches: SH
Screening search results: SH, Elizabeth Steels (ES)
Organizing retrieval of papers: SH
Screening retrieved papers against inclusion criteria: SH, ES
Appraising quality of papers: SH, ES, Anne Chang (AC)
Abstracting data from papers: SH, ES, Kirsten Gibbons (KG)
Writing to authors of papers for additional information: SH
Providing additional data about papers: SH, AC
Obtaining and screening data from unpublished studies: SH, ES
Data management for the review: SH
Entering data into Review Manager (RevMan 5.1): SH, KG
Analysis of data: SH, ES, KG
Interpretation of data: SH, ES, AC, KG
Writing the review: SH, AC, KG
Securing funding for the review: SH
Performing previous work that was the foundation of the present study: SH
Guarantor for the review (one author): SH
Statistical analysis: KG, AC
Sources of support
Internal sources
-
Nursing Research Centre, Mater Health Services, Australia.
Time and facilities.
External sources
-
Queensland Health, Australia.
Nursing and Midwifery Research Grant ($5906) awarded to Sonia Hines
Declarations of interest
Sonia Hines: Queensland Health Nursing and Midwifery Research Grant received by Sonia Hines to assist with the conduct of the review (AUD 5906). The granting body had no influence on the findings of this review.
All other authors: no conflict of interest is known.
Acknowledgements
We thank Mathew Zacharias, Jung T Kim, NL Pace, Peter Kranke and Anne Lyddiatt for their help and advice during the preparation of the systematic review.
We also thank Mathew Zacharias, Katrina Farber, Milli Reddy, Jung T Kim and Janet Wale for their help and editorial advice during the preparation of the protocol for the systematic review.
The authors wish to acknowledge Kathy Hibberd (Librarian, University of Queensland Medical Library) for her invaluable assistance in preparing and conducting the searches for this review, and Leandra Blake for her comments on the protocol and review. We also thank Kate Kynoch and Lisa Brown for assisting with the testing of the data extraction tool.
Thanks to Marie Kristensson for the Swedish translations, Abbas Breesem for the Farsi translation, and Laurie Bay at the Institute of Modern Languages at the University of Queensland for the French translation.
Version history
| Published | Title | Stage | Authors | Version |
| 2018 Mar 10 | Aromatherapy for treatment of postoperative nausea and vomiting | Review | Sonia Hines, Elizabeth Steels, Anne Chang, Kristen Gibbons | |
| 2012 Apr 18 | Aromatherapy for treatment of postoperative nausea and vomiting | Review | Sonia Hines, Elizabeth Steels, Anne Chang, Kristen Gibbons | |
| 2009 Jan 21 | Aromatherapy for treatment of postoperative nausea and vomiting | Protocol | Sonia Hines, Elizabeth Steels, Anne Chang, Kristen Gibbons | |
Differences between protocol and review
The original protocol stated "We will judge the study quality using a validated critical appraisal checklist developed by the Joanna Briggs Institute and based on the work of The Cochrane Collaboration and the Centre for Reviews and Dissemination (Figure 2). This checklist assesses selection, allocation, treatment, and attrition biases". Due to changes in the Cochrane requirements, we have used the Cochrane risk of bias assessment instead.
We had originally planned to search the website http://www.nhmrc.gov.au/nics/asp/index.asp, however this no longer exists and http://www.nhmrc.gov.au/nics/index.htm was searched instead.
Keywords
MeSH
Medical Subject Headings (MeSH) Keywords
- 2‐Propanol [*administration & dosage];
- Administration, Inhalation;
- Antiemetics [*administration & dosage];
- Aromatherapy [*methods];
- Controlled Clinical Trials as Topic;
- Plant Oils [*administration & dosage];
- Postoperative Nausea and Vomiting [*therapy];
- Randomized Controlled Trials as Topic;
- Salvage Therapy [methods];
Medical Subject Headings Check Words
Humans;
PICO

Results of searches
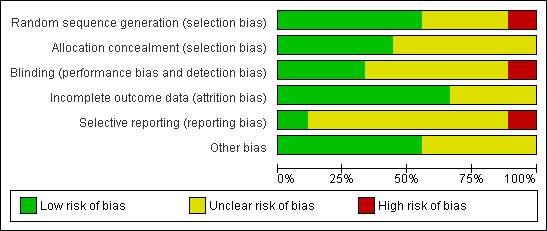
Methodological quality graph: review authors' judgements about each methodological quality item presented as percentages across all included studies.
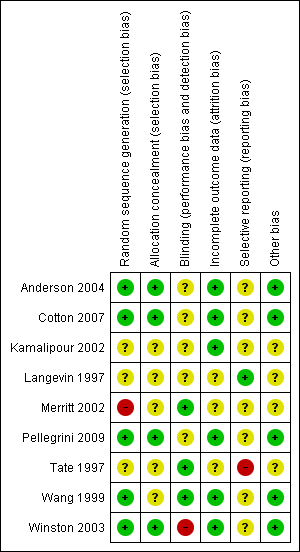
Methodological quality summary: review authors' judgements about each methodological quality item for each included study.
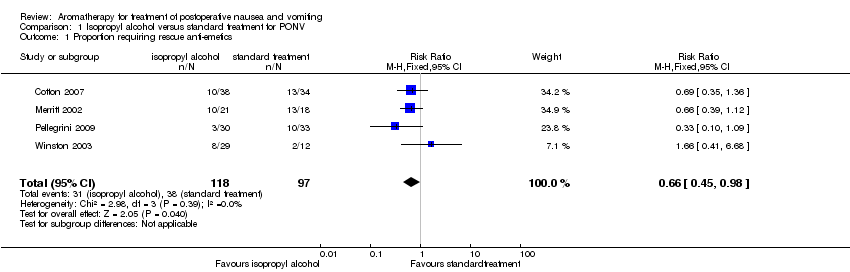
Comparison 1 Isopropyl alcohol versus standard treatment for PONV, Outcome 1 Proportion requiring rescue anti‐emetics.
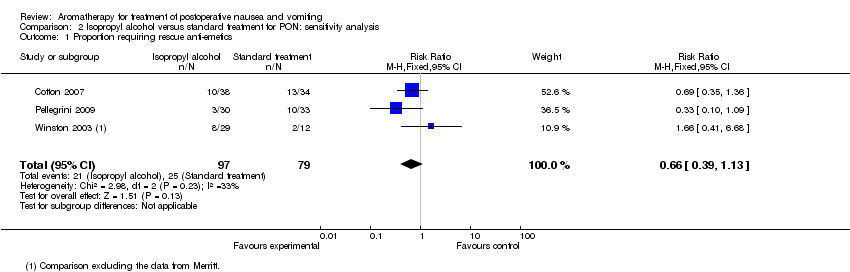
Comparison 2 Isopropyl alcohol versus standard treatment for PON: sensitivity analysis, Outcome 1 Proportion requiring rescue anti‐emetics.

Comparison 3 Isopropyl alcohol versus standard treatment for PON, Outcome 1 Proportion requiring rescue anti‐emetics.

Comparison 4 Isopropyl alcohol versus saline, Outcome 1 Proportion requiring rescue anti‐emetics.
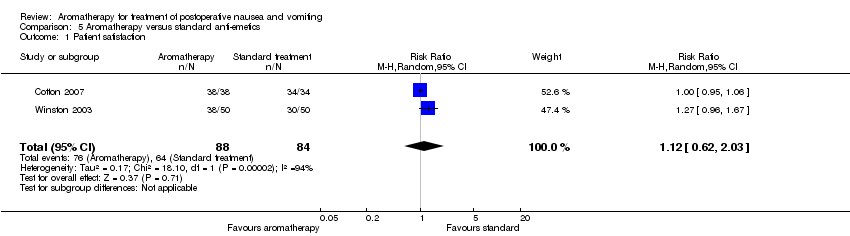
Comparison 5 Aromatherapy versus standard anti‐emetics, Outcome 1 Patient satisfaction.
| Isopropyl alcohol compared to standard treatment for treatment of postoperative nausea and vomiting | ||||||
| Patient or population: patients with treatment of postoperative nausea and vomiting | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Standard treatment | Isopropyl alcohol | |||||
| Requirement for rescue anti‐emetics | Study population1 | RR 0.66 | 215 | ⊕⊕⊝⊝ | ||
| 392 per 1000 | 259 per 1000 | |||||
| Medium risk population1 | ||||||
| 275 per 1000 | 182 per 1000 | |||||
| Adverse effects4 | See comment | See comment | Not estimable | 0 | See comment | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Calculated using control group results. | ||||||
| Isopropyl alcohol compared to saline for treatment of postoperative nausea and vomiting | ||||||
| Patient or population: patients with treatment of postoperative nausea and vomiting | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| saline | Isopropyl alcohol | |||||
| Requirement for rescue anti‐emetics1,2 | Study population3 | RR 0.23 | 135 | ⊕⊕⊝⊝ | ||
| 868 per 1000 | 200 per 1000 | |||||
| Low risk population3 | ||||||
| 100 per 1000 | 23 per 1000 | |||||
| Adverse effects6 | See comment | See comment | Not estimable | 0 | See comment | |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Participants enrolled into study on complaint of nausea and/or vomiting. | ||||||
| Study | Design | Intervention/Control | Outcome | Findings |
| RCT | IPA/ondansetron | Time to 50% reduction in nausea (VNRS1) | IPA: mean 15.00 (SD:10.6mins) Ondansetron: mean 33.88 (SD: 23.2mins) | |
| RCT | IPA/saline | Percentage "response"2 to treatment within 5 minutes | IPA: 78% Saline: 7.3% | |
| CCT | IPA/saline | Percent with complete relief of nausea in 5 minutes | IPA: 80% Saline: 0% | |
| RCT | IPA/Promethazine | Mean time to 50% reduction in nausea scores (VNRS1) | IPA: (mean +/‐ SD) PACU3: 6.43 +/‐ 3.78 minutes SDSU4: 8.33 +/‐ 4.82 minutes HOME5: 16.58 +/‐ 6.9 minutes Promethazine: (mean +/‐ SD) PACU3: 20.5 +/‐ 18.236 minutes SDSU4: 23.3 +/‐ 18.86 minutes HOME5: 26.67 +/‐ 12.5 minutes | |
| RCT | IPA/ondansetron | Mean time to 50% reduction of VNRS1 | IPA: 6.3 minutes Ondansetron: 27.7 minutes | |
| 1VRNS: Verbal Numeric Rating Scale. 2Meaning of response not defined by study authors. 3PACU: Postanaesthesia Care Unit. 4SDSU: Same Day Surgery Unit. 5Home: Participant's residence post‐discharge. | ||||
| Study | Design | Intervention/Control | Outcome | Findings |
| CCT | IPA/standard anti‐emetics | Decrease in mean nausea score (DOS1) 0‐10 (0 = no nausea, 10 = worst nausea and vomiting imaginable) | IPA: Mean DOS1 score Pre‐treatment: 5.71 Post‐treatment: 2.7 Standard treatment: Pre‐treatment: 6.11 Post‐treatment: 1.94 | |
| CCT | Peppermint oil/peppermint essence/standard treatment | Mean daily nausea scores (DOS1) 0‐4 (0 = no nausea, 4 = about to vomit) | Standard treatment: mean daily nausea score = 0.975 Peppermint essence mean daily nausea score (placebo): 1.61 Peppermint oil mean daily nausea score: 0.5 | |
| RCT | IPA/saline | Percentage of participants with decrease in nausea after 3 treatments (VAS) 0‐100 (0 = no nausea, 100 = extreme nausea) | IPA: 91% Saline: 40% | |
| 1DOS: Descriptive Ordinal Scale. | ||||
| Study | Design | Intervention/Comparison | Measure | Satisfied |
| RCT | IPA/ondansetron | 4‐point DOS (poor, fair, good, excellent) | Good or excellent: Intervention: 38/38 Comparison: 34/34 | |
| RCT | IPA/ondansetron | 4‐point DOS (poor, fair, good, excellent) | Good or excellent: Intervention: 38/50 Comparison: 30/50 | |
| RCT | IPA/Promethazine | 5‐point DOS (1 = totally unsatisfied, 5 = totally satisfied) | Both groups report median score 4 | |
| RCT | IPA/Saline/Peppermint | 100mm VAS (0 mm extremely dissatisfied; 100 mm fully satisfied)
| IPA: 90.3 (SD: 14.9) peppermint: 86.3 (SD: 32.3) saline: 83.7 (SD: 25.6)
|
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Proportion requiring rescue anti‐emetics Show forest plot | 4 | 215 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.45, 0.98] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Proportion requiring rescue anti‐emetics Show forest plot | 3 | 176 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.39, 1.13] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Proportion requiring rescue anti‐emetics Show forest plot | 3 | 176 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.39, 1.13] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Proportion requiring rescue anti‐emetics Show forest plot | 3 | 135 | Risk Ratio (M‐H, Random, 95% CI) | 0.30 [0.09, 1.00] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Patient satisfaction Show forest plot | 2 | 172 | Risk Ratio (M‐H, Random, 95% CI) | 1.12 [0.62, 2.03] |

