Limpieza de la herida en las úlceras de decúbito
Referencias
Referencias de los estudios incluidos en esta revisión
Referencias de los estudios excluidos de esta revisión
Referencias adicionales
Referencias de otras versiones publicadas de esta revisión
Characteristics of studies
Characteristics of included studies [ordered by year of study]
Ir a:
| Methods | RCT ‐ single blind. Follow up was for 14 days. Method of allocation unclear. | |
| Participants | In patients with Grade 3 or Grade 4 pressure ulcers (no information on the classification system provided). 18 subjects with 42 ulcers, 18 wounds in non‐whirlpool group and 24 ulcers in whirlpool group. | |
| Interventions | Intervention group: hydrotherapy provided for 20 minutes once a day in a whirlpool bath with water warmed to 96‐98 degrees Fahrenheit. Wounds were irrigated with saline, then dressed with saline‐soaked gauze, dressings changed twice daily. Control group: wounds were irrigated with saline, then dressed with saline‐soaked gauze, dressings changed twice daily. | |
| Outcomes | Improvement in ulcer condition identified by changes in measurement of length and width of the wounds from week to week. Ulcers described as 'improved', 'no change' or 'deteriorated'. Whirlpool group showed superior wound healing (P value = 0.0435), although RevMan analysis did not verify this statistically significant result. | |
| Notes | Information regarding baseline comparability was not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Quote: "All wounds were identified and randomly assigned to one of the two treatment groups, each ulcer was randomised separately". |
| Allocation concealment? | Unclear risk | Not stated. |
| Blinding? | High risk | Not blinded. |
| Blinding? | High risk | Not blinded. |
| Blinding? | Low risk | The outcome assessor was blinded. Quotation: "The grading of wounds was completed by a study physician who was blinded as to the study group". |
| Incomplete outcome data addressed? | Low risk | No withdrawals from the study. The same number of wounds randomised were assessed at 2 weeks. |
| Free of selective reporting? | Low risk | |
| Free of other bias? | Low risk | |
| Methods | RCT, allocation using random numbers table. Follow up for six weeks. | |
| Participants | Patients with chronic wounds of Grade 2 or Grade 3 receiving care in a community setting. | |
| Interventions | Intervention group: (n = 6) ulcers cleansed with tap water. Control group: (n = 2) ulcers cleansed with saline. A combination of hydrocolloid and gel or hydrocolloid alone was used topically on the intervention group, whereas either a hydrocolloid alone or a hydrocolloid and hydrocolloid paste was used in the control group. | |
| Outcomes | Sub group analysis not conducted. Three of the six wounds in the tap water group healed, whereas neither of the two wounds in the saline group healed in the study period. | |
| Notes | This study considered more than one type of wound: i.e. lacerations, venous ulcers and pressure ulcers. Information regarding baseline comparability was not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Low risk | Quote: "using a random numbers table". |
| Allocation concealment? | Unclear risk | Not stated. |
| Blinding? | Unclear risk | Not stated. |
| Blinding? | Low risk | The nurse performing the dressing was blinded. |
| Blinding? | Low risk | Quote: "the project manager was blinded and undertook the assessment of the wound at baseline and at 6 weeks". |
| Incomplete outcome data addressed? | High risk | 8 participants, 4 from each group were "withdrawn because they stopped participating, were admitted to hospital or did not adhere to the treatment". These participants were not included in the analysis. |
| Free of selective reporting? | Low risk | |
| Free of other bias? | Low risk | |
| Methods | Multicentre RCT. Method of allocation unclear. Follow up 14 days. | |
| Participants | Elderly patients of both sexes, with ulcers of > Grade 1 NPUAP scale, dimensions of the ulcer within 10 cm x 10 cm, in patient admission or under home care assistance for greater than 24 hours. | |
| Interventions | Control group: cleansing with isotonic saline solution. | |
| Outcomes | Mean percentage reduction in PSST at day 14: | |
| Notes | Data analysis conducted on 126 participants, seven participants withdrew from the trial (per protocol analysis carried out because it was not possible to assign a PSST score to those withdrawn from the trial). Information regarding baseline comparability was not reported. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Adequate sequence generation? | Unclear risk | Not clearly stated. |
| Allocation concealment? | Unclear risk | Not described. |
| Blinding? | Unclear risk | Not described. |
| Blinding? | Unclear risk | Not described. |
| Blinding? | Unclear risk | Not described. |
| Incomplete outcome data addressed? | Low risk | Seven patients dropped out of the study because they started antibiotic therapy (per protocol analysis carried out). |
| Free of selective reporting? | Low risk | |
| Free of other bias? | Low risk | |
Characteristics of excluded studies [ordered by study ID]
Ir a:
| Study | Reason for exclusion |
| Study explores wound treatment, not wound cleansing | |
| Insufficient information (author contacted but no response received). | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores venous leg ulcers, not pressure ulcers. | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores wound dressings, not wound cleansing. | |
| Not an RCT, study explores wound dressings, not wound cleansing. | |
| Study explores wound dressings, not wound cleansing. | |
| Study explores the prevention of pressure ulcers (i.e. no patient had a pressure ulcer at the start of the study). | |
| Study explores wound treatment, not wound cleansing |
Data and analyses
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Saline versus tap water Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| Analysis 1.1  Comparison 1 Different cleansing solutions, Outcome 1 Saline versus tap water. | ||||
| 1.1 Healed wound within 6 weeks | 1 | 8 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.00 [0.21, 41.89] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Whirlpool versus no whirlpool Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| Analysis 2.1  Comparison 2 Different cleansing techniques, Outcome 1 Whirlpool versus no whirlpool. | ||||
| 1.1 Improvement in pressure ulcer | 1 | 42 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.1 [0.93, 4.76] |

Methodological quality summary: review authors' judgements about each methodological quality item for each included study.

Comparison 1 Different cleansing solutions, Outcome 1 Saline versus tap water.
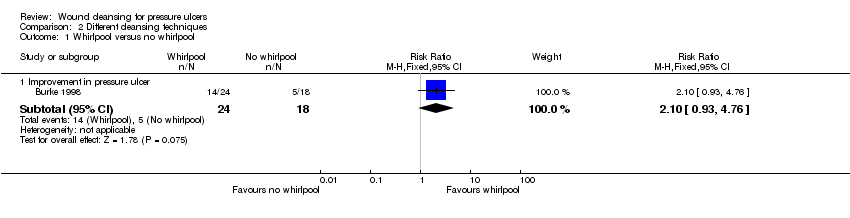
Comparison 2 Different cleansing techniques, Outcome 1 Whirlpool versus no whirlpool.
| Intervention | PSST Baseline | PSST Day 7 | PSST Day 14 | Total % Change |
| Isotonic saline solution (control) | mean 31.6 (SD 10.3, min 15.0, max 52.0) | 28.9 (SD 10.5, min 12.0, max 52.0). | 25.3 (SD 12.2, min 10.0, max 50.0). | ‐20.5 (SD 24.1, min ‐65.8, max 22.7). |
| Saline spray, Aloe vera, silver chloride and decyl glucoside (Vulnopur) (intervention) | mean 31.3 (SD 11.5, min 13.0 max 56.0) | 27.1 (SD 11.1, min 13.0, max 54.0). | 21.6 (SD 11.6, min 10.0, max 51,0). | ‐27.8 (SD 31.3, min ‐69.8, max 123.5). |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Saline versus tap water Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Healed wound within 6 weeks | 1 | 8 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.00 [0.21, 41.89] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Whirlpool versus no whirlpool Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Improvement in pressure ulcer | 1 | 42 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.1 [0.93, 4.76] |

