Balón intragástrico para la obesidad
Referencias
Referencias de los estudios incluidos en esta revisión
Referencias de los estudios excluidos de esta revisión
Referencias adicionales
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Ir a:
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 12 weeks: | |
| Outcomes | Weight change (%of loss or kg): Weight change (BMI): B+S x S+B P = 0.3 Last 12 weeks: B+S x S+B P = 0.3 Losses: Complications: | |
| Notes | In 24 weeks of study: | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 4 groups | |
| Outcomes | Weight change (%of loss or kg): Losses: 16 | |
| Notes | First months: | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Methods | Design: | |
| Participants | Country: Obese indivuduals above ideal weight | |
| Interventions | 3 months with balloon + 9 months of follow‐up | |
| Outcomes | 56 patients completed the first 3 months: 22 sham + 34 balloon Weight change (%of loss or kg): Balloon = ‐ 7.2% Weight change (BMI): Normal balloons or over 70% full: Losses: Side‐effects: | |
| Notes | The loss of weight in the follow‐up was difficult to evaluate due to the high rate of drop‐outs after removing the balloon; the patients in the final meeting (28 balloon, 16 sham) showed a continuous tendency of weight loss in the control group and of weight gain in the balloon treated group; | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Methods | Design: | |
| Participants | Country: 21%‐77% above ideal body weight: | |
| Interventions | 11 underwent insertion of intragastric balloon and 11 underwent sham procedures. | |
| Outcomes | 8 in 10 balloons were found deflated. Baloon group: 1 patient showed migration of a deflated balloon through all the intestinal tract; 1 patient had abdominal pain, withdrawing only 3 days after balloon insertion. Sham group: 2 patients with gastric erosions associated with the use of aspirin. | |
| Notes | There was no important difference in weight loss between these 2 groups. Costs: The balloon and introducer costs were approximately $400 and the endoscopic charges for insertion and removal of each balloon cost $1200 to $1600 in addition. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 4 Groups | |
| Outcomes | Weight change (%of loss or kg) of all patients: BMI change of all patients: Side‐effects in balloon patients: Hiccups and gastric fullness. Side‐effects in sham‐treated patients: nausea, heartburn and belching; 36% were sure that they felt a balloon after the sham Losses: | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 3m group 1: Sham 3‐6m group 1: Balloon After 6months until year one: removal of all balloons and observation of weight maintenance | |
| Outcomes | 10 excluded patients: Weight change (%of loss or kg): 3‐6 months: After 6 months, the overall weight loss was: Weight loss in 33 patients who had completed the study per protocol: 47% of all patients sustained a greater than 10% weight loss, with considerably reduced comorbidity. Complications: Gastric erosions, gastric ulcers, minor gastric bleeding, Mallory‐Weiss tears, oesophageal lacerations | |
| Notes | 55% of patients maintained a weight loss greater than 10%. Although an independent benefit of balloon treatment beyond diet, exercise, and behavioural therapy could not be demonstrated in the first 3 months, balloon treatment for 1 year resulted in a substantial weigh loss, the greater part of which was maintained during the balloon‐free second year. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 12 weeks balloon + 12 weeks sham 12 weeks sham + 12 weeks balloon | |
| Outcomes | Weight change (%of loss or kg): sham +‐ 0.8 kg (14 patients) BMI change: Losses: *1 migration of deflated balloon through abdominal tract Side‐effects: | |
| Notes | Neither mean cumulative weight reduction nor BMI in the 2 evaluation periods (12 wks each) differed significantly; | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | Balloon in 12 patients | |
| Outcomes | Weight change (%of loss or kg): Balloon ‐7.33 kg 8 losses: 5 put on weight, 2 did not lose weight, did not attend follow‐ up Side‐effects: | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
| Methods | Design: | |
| Participants | Country: | |
| Interventions | 11 received air‐filled balloons, | |
| Outcomes | Weight change (%of loss or kg): Balloon ‐8.6 kg Weight change (BMI): Side‐Effects "sensation of balloon presence", "gastric distension", "hunger sensation". There was a considerable correlation between the distension and the presence / sensation of the balloon (r=0.901, p<0.001) and a negative correlation between the hunger and the sensation of distension (r=0.8, p<0.0001) | |
| Notes | The cumulative loss was not significantly different between two groups. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Characteristics of excluded studies [ordered by study ID]
Ir a:
| Study | Reason for exclusion |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Inappropriate method of randomisation | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Intervention: comparison of the use of anti‐emetics after intragastric balloon insertion | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Not a randomised controlled trial | |
| Inappropriate method of randomisation | |
| Not a randomised controlled trial |
Data and analyses
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 2 | 61 | Mean Difference (IV, Fixed, 95% CI) | 3.49 [2.75, 4.23] |
| Analysis 1.1  Comparison 1 Intragastric balloon versus diet, Outcome 1 Weight loss in kg. | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| Analysis 2.1  Comparison 2 Intragastric balloon versus no treatment, Outcome 1 Weight loss in kg. | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| Analysis 3.1  Comparison 3 Intragastric balloon versus intragastric balloon + diet, Outcome 1 Weight loss in kg. | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 5 | 158 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.44, 0.35] |
| Analysis 4.1  Comparison 4 Intragastric balloon + diet versus diet only, Outcome 1 Weight loss in kg. | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Gastric ulcers Show forest plot | 6 | 449 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.91 [1.57, 15.35] |
| Analysis 5.1  Comparison 5 Minor complications due to balloon usage, Outcome 1 Gastric ulcers. | ||||
| 2 Gastric erosions Show forest plot | 6 | 489 | Risk Ratio (M‐H, Fixed, 95% CI) | 9.78 [3.87, 24.69] |
| Analysis 5.2  Comparison 5 Minor complications due to balloon usage, Outcome 2 Gastric erosions. | ||||
| 3 Abdominal pain Show forest plot | 4 | 272 | Risk Ratio (M‐H, Fixed, 95% CI) | 14.0 [3.45, 56.74] |
| Analysis 5.3  Comparison 5 Minor complications due to balloon usage, Outcome 3 Abdominal pain. | ||||
| 4 Vomiting Show forest plot | 2 | 196 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.33 [0.81, 23.21] |
| Analysis 5.4  Comparison 5 Minor complications due to balloon usage, Outcome 4 Vomiting. | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Migration of deflated balloon Show forest plot | 2 | 54 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.67 [0.30, 24.01] |
| Analysis 6.1  Comparison 6 Major complications due to balloon usage, Outcome 1 Migration of deflated balloon. | ||||
| 2 Small bowel obstruction Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| Analysis 6.2  Comparison 6 Major complications due to balloon usage, Outcome 2 Small bowel obstruction. | ||||
| 3 Mallory‐Weiss tears Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| Analysis 6.3  Comparison 6 Major complications due to balloon usage, Outcome 3 Mallory‐Weiss tears. | ||||
| 4 Oesophageal laceration Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| Analysis 6.4  Comparison 6 Major complications due to balloon usage, Outcome 4 Oesophageal laceration. | ||||

Comparison 1 Intragastric balloon versus diet, Outcome 1 Weight loss in kg.
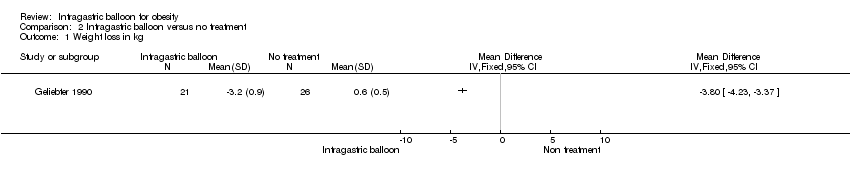
Comparison 2 Intragastric balloon versus no treatment, Outcome 1 Weight loss in kg.
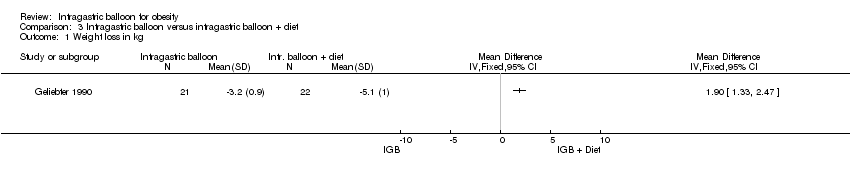
Comparison 3 Intragastric balloon versus intragastric balloon + diet, Outcome 1 Weight loss in kg.

Comparison 4 Intragastric balloon + diet versus diet only, Outcome 1 Weight loss in kg.
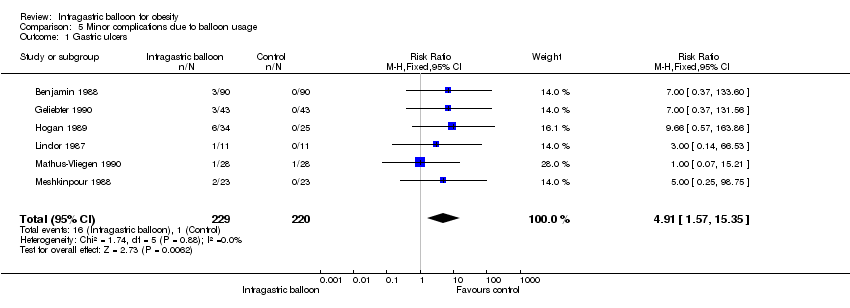
Comparison 5 Minor complications due to balloon usage, Outcome 1 Gastric ulcers.
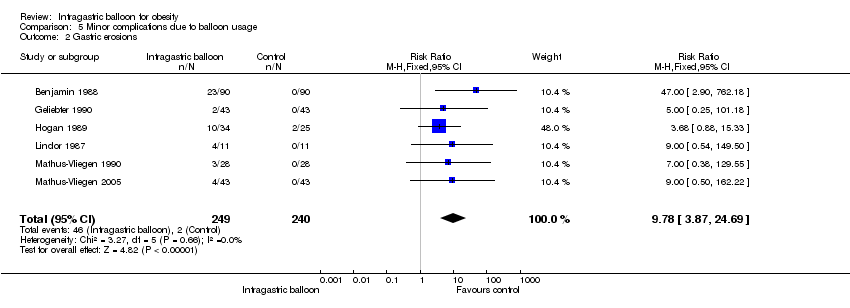
Comparison 5 Minor complications due to balloon usage, Outcome 2 Gastric erosions.

Comparison 5 Minor complications due to balloon usage, Outcome 3 Abdominal pain.
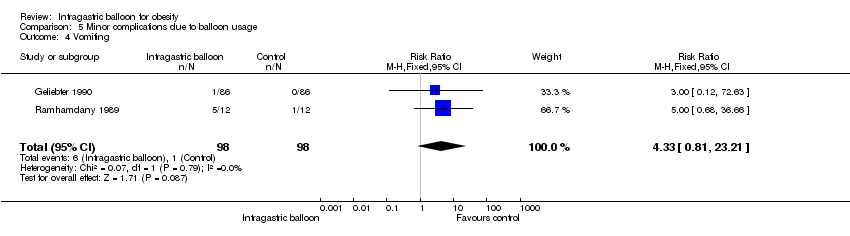
Comparison 5 Minor complications due to balloon usage, Outcome 4 Vomiting.
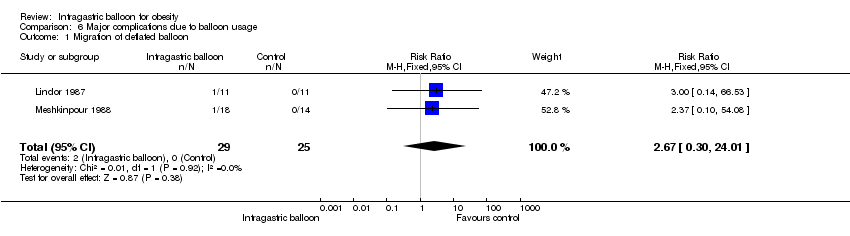
Comparison 6 Major complications due to balloon usage, Outcome 1 Migration of deflated balloon.
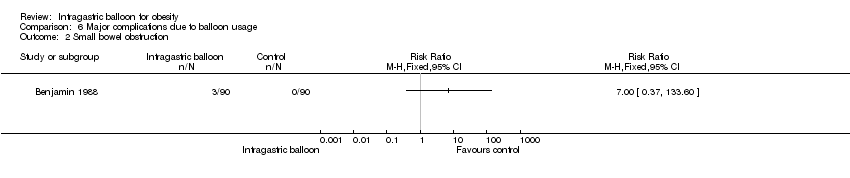
Comparison 6 Major complications due to balloon usage, Outcome 2 Small bowel obstruction.
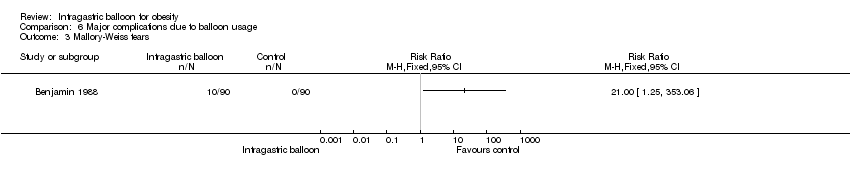
Comparison 6 Major complications due to balloon usage, Outcome 3 Mallory‐Weiss tears.
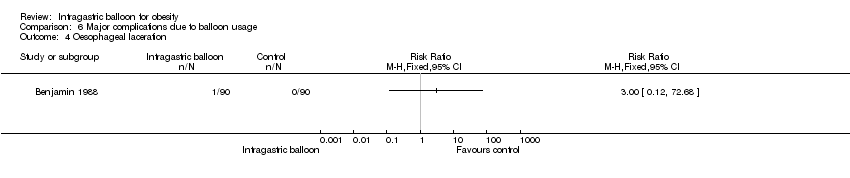
Comparison 6 Major complications due to balloon usage, Outcome 4 Oesophageal laceration.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 2 | 61 | Mean Difference (IV, Fixed, 95% CI) | 3.49 [2.75, 4.23] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Weight loss in kg Show forest plot | 5 | 158 | Mean Difference (IV, Fixed, 95% CI) | ‐0.04 [‐0.44, 0.35] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Gastric ulcers Show forest plot | 6 | 449 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.91 [1.57, 15.35] |
| 2 Gastric erosions Show forest plot | 6 | 489 | Risk Ratio (M‐H, Fixed, 95% CI) | 9.78 [3.87, 24.69] |
| 3 Abdominal pain Show forest plot | 4 | 272 | Risk Ratio (M‐H, Fixed, 95% CI) | 14.0 [3.45, 56.74] |
| 4 Vomiting Show forest plot | 2 | 196 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.33 [0.81, 23.21] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Migration of deflated balloon Show forest plot | 2 | 54 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.67 [0.30, 24.01] |
| 2 Small bowel obstruction Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 3 Mallory‐Weiss tears Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4 Oesophageal laceration Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |

