Amnio‐infusion en cas de rupture prématurée des membranes avant terme au troisième trimestre
Résumé scientifique
Contexte
La rupture prématurée des membranes avant terme (RPMAT) est l’une des principales causes de morbidité et de mortalité périnatale. L’amnio‐infusion consiste à rétablir le volume de liquide amniotique en introduisant une solution dans la cavité utérine.
Objectifs
L’objectif de cette revue était d’évaluer les effets de l’amnio‐infusion sur la morbidité et la mortalité périnatale et maternelle en cas de RPMAT.
Stratégie de recherche documentaire
Nous avons effectué une recherche dans le registre d’essais cliniques du groupe Cochrane sur la grossesse et l’accouchement (2 décembre 2013).
Critères de sélection
Essais randomisés portant sur l’amnio‐infusion comparés à l’absence d’amnio‐infusion chez les femmes ayant subi une RPMAT.
Recueil et analyse des données
Trois auteurs de la revue ont évalué les essais à inclure de façon indépendante. Deux auteurs de revue ont évalué la qualité des essais et extrait les données de manière indépendante. L’exactitude des données a été vérifiée.
Résultats principaux
Nous avons inclus cinq essais, de qualité modérée, mais seules les données issues de quatre études (avec un total de 241 participantes) ont été analysées. Un essai n’a fourni aucune des données de la revue.
L’amnio‐infusion transcervicale a amélioré le pH de l’artère ombilicale fœtale au moment de l’accouchement (différence moyenne 0,11 ; intervalle de confiance (IC) à 95 % 0,08 à 0,14 ; un essai, 61 participantes) et réduit les décélérations variables persistantes pendant l’accouchement (risque relatif (RR) 0,52 ; IC à 95 % 0,30 à 0,91 ; un essai, 86 participantes).
L’amnio‐infusion trans‐abdominale était associée à une baisse de la mort néonatale (RR 0,30 ; IC à 95 % 0,14 à 0,66 ; deux essais, 94 participantes), du sepsis néonatal (RR 0,26 ; IC à 95 % 0,11 à 0,61 ; un essai, 60 participantes), de l’hypoplasie pulmonaire (RR 0,22 ; IC à 95 % 0,06 à 0,88 ; un essai, 34 participantes) et du sepsis puerpéral (RR 0,20 ; IC à 95 % 0,05 à 0,84 ; un essai, 60 participantes). La propension des femmes appartenant au groupe d’amnio‐infusion à accoucher dans les sept jours suivant la rupture de la membrane (RR 0,18 ; IC à 95 % 0,05 à 0,70 ; un essai, 34 participantes) était également en baisse. Ces résultats doivent être considérés avec circonspection car ces découvertes positives provenaient principalement d’un seul essai avec une assignation secrète imprécise.
Conclusions des auteurs
Ces résultats sont encourageants, mais ne se limitent qu’à quelques données et une robustesse méthodologique confuse. Par conséquent, d’autres preuves doivent être établies que l’amnio‐infusion en cas de RPMAT puisse être recommandée dans le cadre d’une pratique clinique systématique.
PICO
Résumé simplifié
Amnio‐infusion en cas de rupture prématurée des membranes avant terme
Il existe des preuves démontrant que le rétablissement du volume de liquide amniotique à l’aide d’une solution saline ou d’un liquide semblable (amnio‐infusion) après la rupture prématurée des membranes avant terme (RPMAT) peut se révéler bénéfique pour les bébés nés avant terme (en prévenant les risques d’infections, de lésions pulmonaires et de mort) et les mères (en prévenant les risques d’infections utérines après l’accouchement). Toutefois, les preuves actuelles sont insuffisantes pour recommander le recours systématique à une amnio‐infusion en cas de RPMAT.
La rupture prématurée des membranes avant terme est la seule cause d’accouchement prématuré la plus identifiable. Le sac (membranes) entourant le bébé et le liquide utérin se rompent (ruptures) généralement pendant l’accouchement. Si les membranes se rompent avant l’accouchement et avant le terme (avant 37 semaines), le bébé est exposé à un risque élevé d’infection. Une diminution du volume de liquide entourant le bébé augmente également les risques de compression du cordon ombilical, réduisant ainsi l’apport en nutriments et en oxygène du bébé. De plus, une insuffisance de liquide amniotique peut altérer le développement pulmonaire normal des bébés et entraîner des souffrances fœtales avec des variations du rythme cardiaque. Un surplus de liquide peut être injecté dans le vagin de la mère (amnio‐infusion transcervicale) ou via l’abdomen (amnio‐infusion transabdominale) dans l’utérus, afin d’ajouter davantage de liquide pour le bébé. La revue composée de cinq essais contrôlés randomisés (contenant des données issues d’analyses effectuées auprès de 241 participantes) a trouvé des preuves démontrant que l’amnio‐infusion réalisée avec une solution saline peut améliorer les résultats sur la santé et se révèle bénéfique pour les bébés et les mères après une RPMAT. Toutefois, ces preuves sont actuellement insuffisantes pour recommander son recours systématique en raison du nombre limité d’essais et d’un faible nombre de femmes y participant.
Authors' conclusions
Summary of findings
| Transabdominal amnioinfusion compared with no amnioinfusion for preterm rupture of membranes (PROM) | ||||
| Patient or population: pregnant women with PROM Settings: hospital Intervention: transabdominal amnioinfusion Comparison: no amnioinfusion | ||||
| Outcomes | Relative effect | No of Participants | Quality of the evidence | Comments |
| Neonatal death | RR 0.30 (0.14 ‐ 0.66) | 94 (two studies) | ⊕⊕⊕⊝ | Risk of neonatal death in the amnioinfusion group was 127 per 1000 compared to 426 per 1000 in the control group. |
| Neonatal sepsis/infection | RR 0.26 (0.11 ‐ 0.61) | 60 (one study*) | ⊕⊕⊕⊝ | *Sepsis defined as micro‐erythrocyte sedimentation rate > 5 mm, total leucocyte count < 5000, CRP > 6 mg/dL, platelet count < 100,000 or a positive blood culture within the first 48 hours. |
| Pulmonary hypoplasia | RR 0.22 (0.06 ‐ 0.88) | 34 (one study) | ⊕⊕⊝⊝ | Pulmonary hypoplasia was diagnosed according to strict clinical and radiological criteria, however, this study was small and blinding to group allocation was not described and so we downgraded this evidence from moderate to low. More evidence is needed. |
| Maternal puerperal sepsis | RR 0.20 (0.05 ‐ 0.84) | 60 (one study**) | ⊕⊕⊕⊝ | **Defined as fever > 38° C and a positive high vaginal swab culture. |
| GRADE Working Group grades of evidence CI: confidence interval; CRP: C‐reactive protein; RR: risk ratio | ||||
Background
Description of the condition
Preterm premature rupture of membranes (PPROM) remains the single most identifiable cause of preterm labour and a major contributor to perinatal mortality and morbidity. Oligohydramnios (reduced amniotic fluid volume) following PPROM is associated with a higher risk of chorioamnionitis, neonatal fetal infection and cord compression (Keirse 1989; Vintzileos 1985). Umbilical cord compression may cause persistent variable fetal heart rate decelerations (Gabbe 1976). Oligohydramnios is also the most important predictor of perinatal mortality in very early PPROM and adequate residual amniotic fluid plays a critical role in determining the prevalence of pulmonary hypoplasia, which is a major cause of death in these babies (Vergani 1994; Vintzileos 1985). Abnormal neurological outcomes and postural deformities in the neonate may also occur as a consequence of PPROM (Locatelli 2000).
Description of the intervention
Saline fluid or Ringers lactate/Hartmans is infused transcervically through a catheter into the uterine cavity, or transabdominally, through a narrow gauge needle. Amnioinfusion was first described as a method of preventing or relieving umbilical cord compression during labour (Miyazaki 1983). The technique has since been used prophylactically in various conditions associated with oligohydramnios, including impaired intrauterine growth and PPROM. Amnioinfusion has been shown to prolong the latency period in second trimester PPROM (Locatelli 2000; Ogunyemi 2002; Turhan 2002), improve perinatal survival (Locatelli 2006; Ogunyemi 2002) and decrease rates of pulmonary hypoplasia (Locatelli 2000). Combined with antibiotics and without, it has been used to treat and to prevent infection following premature rupture of membranes (Goodlin 1981; Monahan 1995; Ogita 1988). In a Cochrane review on amnioinfusion for suspected or potential cord compression in labour at term, amnioinfusion improved short‐term measures of neonatal outcome, reduced the use of caesarean section and reduced maternal puerperal sepsis (Hofmeyr 2012). A subcutaneously implanted amniotic fluid replacement port system has been developed for long‐term amnioinfusion in women with preterm premature rupture of the membranes (Tchirikov 2010).
How the intervention might work
Restoring the amniotic fluid volume cushions the fetus, thereby preventing mechanical compression of the umbilical cord and reducing fetal distress. It may prevent fetal lung hypoplasia in PPROM by preventing mechanical compression of the fetal thorax and enabling normal amniotic fluid flow into the fetal lungs (Tranquilli 2005). This effect occurs mainly in the second trimester and is considered in a separate review (Van Teeffelen 2013). Likewise, by preventing mechanical compression of the fetus, amnioinfusion may prevent postural deformities. In addition, the infused fluid may prevent intrauterine infection, possibly by the anti‐bacterial effect of saline, or a diluent effect. Improvements in fetal ductus venosus and umbilical artery flow have been demonstrated following amnioinfusion for PPROM (Hsu 2009).
Why it is important to do this review
Evidence from cohort studies suggests that replenishing the amniotic fluid volume in pregnancies complicated by PPROM is beneficial to mother and child. However, amnioinfusion is an invasive procedure and not without potential risks, thus a thorough evaluation of randomised controlled trials is required.
['Amnioinfusion for cord compression' (Hofmeyr 2012), 'Prophylactic versus therapeutic amnioinfusion' (Novikova 2012), and 'Amnioinfusion for meconium‐stained liquor' (Hofmeyr 2014), are separate reviews.]
Objectives
To assess from the best available evidence the effects of amnioinfusion for preterm premature rupture of membranes on perinatal morbidity and mortality.
Methods
Criteria for considering studies for this review
Types of studies
Clinical trials comparing the effect of amnioinfusion for third trimester preterm premature rupture of the membranes before 37 weeks with a control group (no amnioinfusion); random allocation to treatment and control groups, with adequate allocation concealment; violations of allocated management and exclusions after allocation not sufficient to materially affect outcomes. Quasi‐randomised (alternate allocation), cross‐over and cluster trials were not considered eligible for inclusion.
Types of participants
Pregnant women with preterm premature rupture of membranes.
Types of interventions
Amnioinfusion compared with no amnioinfusion.
Types of outcome measures
Primary outcomes
-
Indicators of fetal condition, e.g. persistent variable decelerations, Apgar scores, cord arterial pH at birth.
-
Neonatal morbidity including infection, lung hypoplasia, abnormal neurological outcomes and postural deformities.
-
Perinatal mortality.
Secondary outcomes
-
Mode of delivery.
-
Indications for delivery.
-
Latency period from amnioinfusion to delivery.
-
Maternal morbidity, e.g. endometritis, postpartum temperature greater than 38°C.
-
Birthweight.
-
Admission to neonatal intensive or high care unit.
We considered outcomes separately for transcervical and transabdominal amnioinfusion.
The methods section of this review is based on a standard template used by the Cochrane Pregnancy and Childbirth Group.
Search methods for identification of studies
Electronic searches
We searched the Cochrane Pregnancy and Childbirth Group’s Trials Register by contacting the Trials Search Co‐ordinator (2 December 2013).
The Cochrane Pregnancy and Childbirth Group’s Trials Register is maintained by the Trials Search Co‐ordinator and contains trials identified from:
-
monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL);
-
weekly searches of MEDLINE;
-
monthly searches of Embase;
-
handsearches of 30 journals and the proceedings of major conferences;
-
weekly current awareness alerts for a further 44 journals plus monthly BioMed Central email alerts.
Details of the search strategies for CENTRAL, MEDLINE and Embase, the list of handsearched journals and conference proceedings, and the list of journals reviewed via the current awareness service can be found in the ‘Specialized Register’ section within the editorial information about the Cochrane Pregnancy and Childbirth Group.
Trials identified through the searching activities described above are each assigned to a review topic (or topics). The Trials Search Co‐ordinator searches the register for each review using the topic list rather than keywords.
We did not apply any language restrictions.
Data collection and analysis
No new studies have been included for this update (2014).
For methods used in the previous version of this review, please see Hofmeyr 2011.
For methods to be used in the next update, see Appendix 1.
Selection of studies
T Lawrie (TL), AC Eke (ACE) and GJ Hofmeyr (GJH) independently assessed for inclusion all potential studies. No studies were identified from the updated search. Two studies previously in ongoing were excluded (Roberts: AMIPROM; Vergani 2007). We resolved any disagreement through discussion. We evaluated trials under consideration for methodological quality and appropriateness for inclusion according to the prespecified selection criteria, without consideration of their results.
Results
Description of studies
In total, we have included five studies; three in the transcervical amnioinfusion comparison (Gonzalez 2001; Nageotte 1985; Puertas 2007) and two in the transabdominal comparison (Singla 2010; Tranquilli 2005). One included study contributed no data (Gonzalez 2001). SeeCharacteristics of included studies. The gestational age range for recruitment to the transcervical trials was 26 to 36+ weeks and in the transabdominal trials, 24 to 34 weeks. Transcervical amnioinfusion was performed in a total of 72 women versus 75 controls (excluding unusable data from Gonzalez 2001) and transabdominal amnioinfusion was performed in 47 women versus 47 controls; thus participant numbers for this review are small.
Risk of bias in included studies
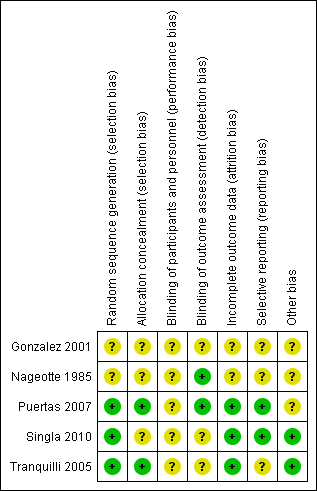
All included studies were randomised trials. SeeFigure 1 and Figure 2 for a summary of 'Risk of bias' assessments.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.

'Risk of bias' graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
Allocation
Randomisation was by random number table in Puertas 2007 and was computer‐generated in Tranquilli 2005 and Singla 2010; the method of randomisation and group allocation was not described in Nageotte 1985 and Gonzalez 2001. Allocation concealment was described in two trials (Puertas 2007; Tranquilli 2005).
Blinding
Partial blinding was described in two trials (Nageotte 1985; Puertas 2007).
Incomplete outcome data
Incomplete outcome data were noted in the randomised clinical trial by Gonzalez et al. This is because it is an abstract only publication, hence provided insufficient detail for assessment (Gonzalez 2001). Five post‐randomisation exclusions: three for non‐vertex presentation and two for fetal distress were noted in the randomised trial by Nageotte et al (Nageotte 1985). Otherwise, outcome data were complete for Puertas 2007, Singla 2010 and Tranquilli 2005.
Selective reporting
In the randomised clinical trial by Tranquilli et al, the authors did not report the mode of delivery, Apgar scores or cord arterial pH for the babies delivered (Tranquilli 2005). No selective reporting was found in the Puertas 2007 and Singla 2010 trials. There was insufficient information to assess selective reporting in Gonzalez 2001 and Nageotte 1985.
Other potential sources of bias
Another source of bias was noted in the randomised clinical trial by Puertas et al, where more carriers of group B streptococcal were noted in control group (14 women versus seven women) (Puertas 2007). Even though all these women with Group B streptococcal infection received prophylactic intrapartum antibiotics, the higher incidence of Group B Streptococcus in the control group was a source of bias for the trial. No other biases were reported for Singla 2010 and Tranquilli 2005. There was insufficient information to assess other sources of bias in Gonzalez 2001 and Nageotte 1985.
Effects of interventions
See: Summary of findings for the main comparison
1. Transcervical amnioinfusion
Transcervical amnioinfusion improved fetal umbilical artery pH at delivery (mean difference (MD) 0.11; 95% confidence interval (CI) 0.08 to 0.14; one trial, 61 participants; Analysis 1.6) and reduced persistent variable decelerations during labour (risk ratio (RR) 0.52; 95% CI 0.30 to 0.91; one trial, 86 participants; Analysis 1.1). No significant differences in the rates of caesarean section, low Apgar scores, neonatal death or infectious morbidity were detected.
2. Transabdominal amnioinfusion
Transabdominal amnioinfusion was associated with a reduction in neonatal death (RR 0.30; 95% CI 0.14 to 0.66; two trials, 94 women; Analysis 2.7), neonatal infection/sepsis (RR 0.26; 95% CI 0.11 to 0.61; one trial, 60 participants; Analysis 2.3), and pulmonary hypoplasia (RR 0.22; 95% CI 0.06 to 0.88; one trial, 34 participants; Analysis 2.3). Women in the amnioinfusion group were also less likely to deliver within seven days of membrane rupture (RR 0.18; 95% CI 0.05 to 0.70; one trial, 34 participants; Analysis 2.4) and were less likely to experience puerperal sepsis (RR 0.20; 95% CI 0.05 to 0.84; one trial, 60 participants; Analysis 2.9). There were no significant differences between groups regarding birthweight, gestational age at delivery, admission to neonatal intensive care and neurological sequelae. Fetal distress was diagnosed less frequently in the amnioinfusion group than the control group (RR 0.27; 95% CI 0.08 to 0.88; one trial, 60 participants; Analysis 2.1), but in the absence of blinding this outcome is at risk of bias.
Discussion
Summary of main results
Transabdominal amnioinfusion following preterm premature rupture of the membranes (PPROM) resulted in better neonatal outcomes (decreased sepsis, infection and death) and decreased puerperal sepsis than conventional management. The data contributing to these outcomes were mainly from one trial (Singla 2010). Transcervical amnioinfusion was associated with improved fetal heart rate patterns and umbilical cord blood pH results, but improvements in substantive clinical outcomes were not statistically significant.
Overall completeness and applicability of evidence
These results are not conclusive due to the small numbers of trials and participants. In addition, the numbers are too small to detect rare potential adverse events. The lower threshold of gestational age at which amnioinfusion may be of benefit is not known.
Quality of the evidence
The evidence is of mainly moderate quality (summary of findings Table for the main comparison) and limitations can largely be attributed to the small size of studies and the lack of blinding to group allocation.
Agreements and disagreements with other studies or reviews
This evidence supports the positive findings of the non‐randomised studies conducted to date.
'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.

'Risk of bias' graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
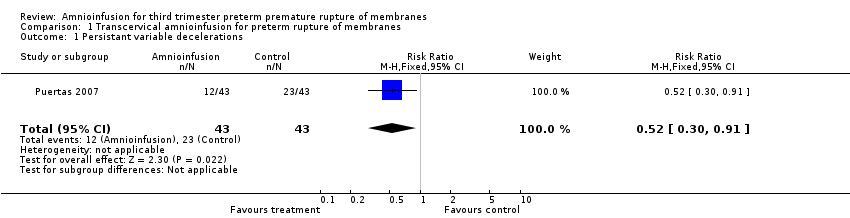
Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 1 Persistant variable decelerations.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 2 Severe variable decelerations per hour in first stage.
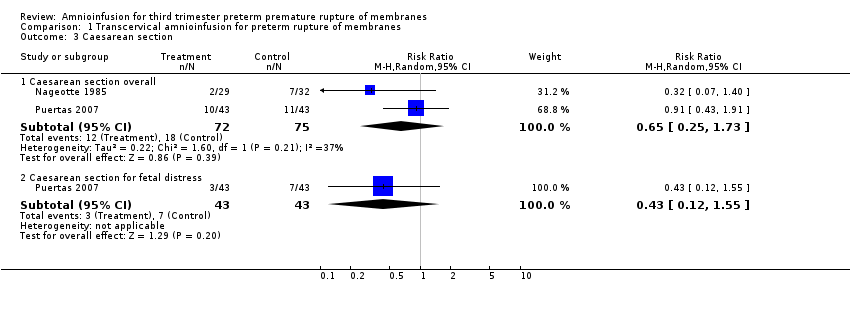
Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 3 Caesarean section.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 4 Forceps/vacuum assisted delivery.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 5 1 minute Apgar score < 4.
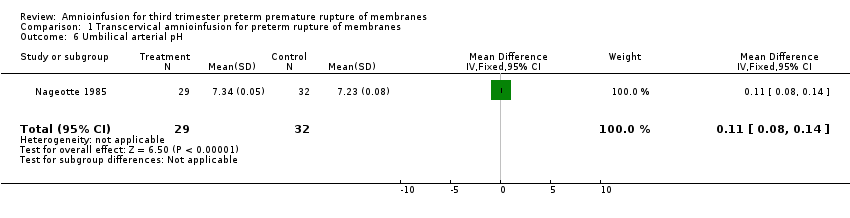
Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 6 Umbilical arterial pH.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 7 Umbilical pH ≤ 7.20.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 8 Neonatal morbidity.

Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 9 Neonatal death.
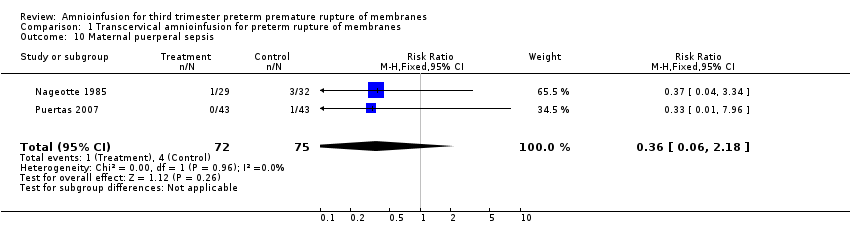
Comparison 1 Transcervical amnioinfusion for preterm rupture of membranes, Outcome 10 Maternal puerperal sepsis.
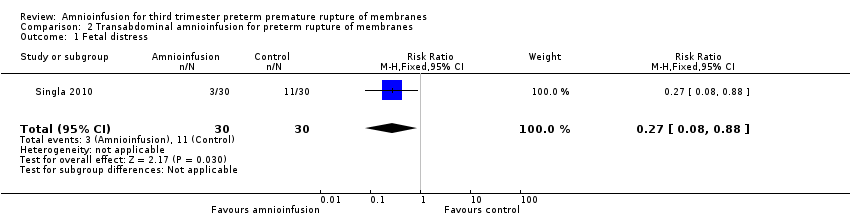
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 1 Fetal distress.

Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 2 Gestational age at delivery (weeks).
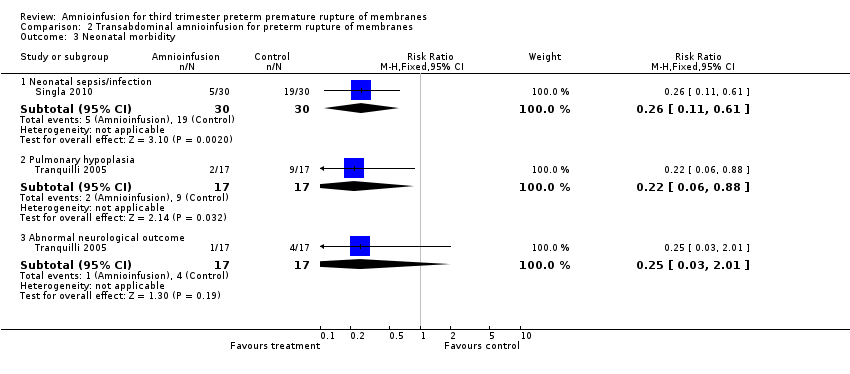
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 3 Neonatal morbidity.
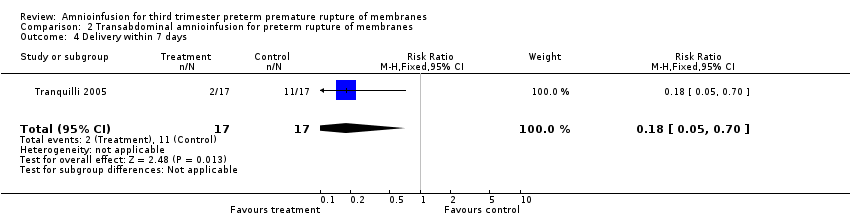
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 4 Delivery within 7 days.

Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 5 Time to delivery (days).

Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 6 Admission to neonatal intensive care unit.
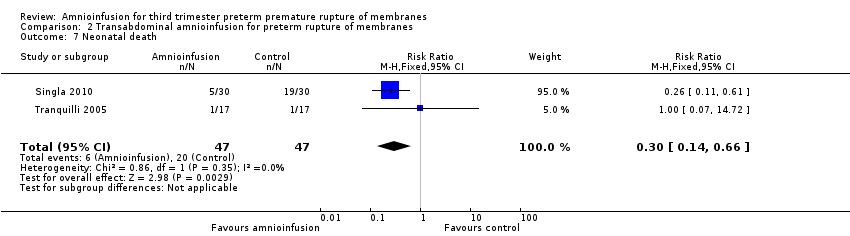
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 7 Neonatal death.
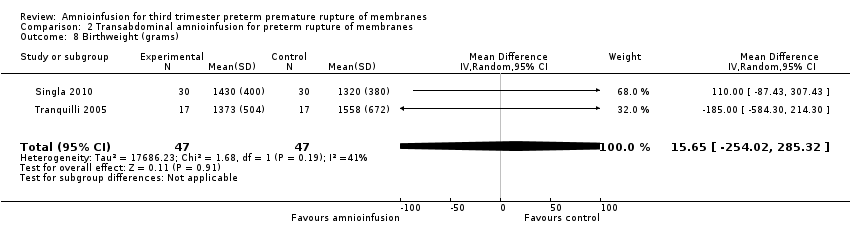
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 8 Birthweight (grams).
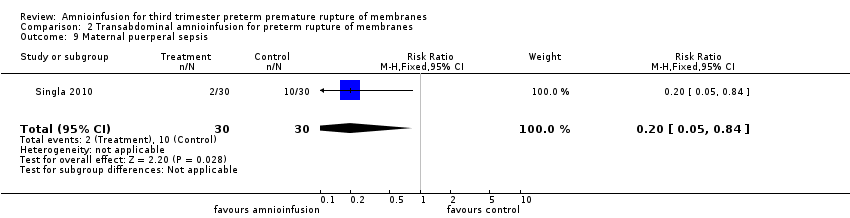
Comparison 2 Transabdominal amnioinfusion for preterm rupture of membranes, Outcome 9 Maternal puerperal sepsis.
| Transabdominal amnioinfusion compared with no amnioinfusion for preterm rupture of membranes (PROM) | ||||
| Patient or population: pregnant women with PROM Settings: hospital Intervention: transabdominal amnioinfusion Comparison: no amnioinfusion | ||||
| Outcomes | Relative effect | No of Participants | Quality of the evidence | Comments |
| Neonatal death | RR 0.30 (0.14 ‐ 0.66) | 94 (two studies) | ⊕⊕⊕⊝ | Risk of neonatal death in the amnioinfusion group was 127 per 1000 compared to 426 per 1000 in the control group. |
| Neonatal sepsis/infection | RR 0.26 (0.11 ‐ 0.61) | 60 (one study*) | ⊕⊕⊕⊝ | *Sepsis defined as micro‐erythrocyte sedimentation rate > 5 mm, total leucocyte count < 5000, CRP > 6 mg/dL, platelet count < 100,000 or a positive blood culture within the first 48 hours. |
| Pulmonary hypoplasia | RR 0.22 (0.06 ‐ 0.88) | 34 (one study) | ⊕⊕⊝⊝ | Pulmonary hypoplasia was diagnosed according to strict clinical and radiological criteria, however, this study was small and blinding to group allocation was not described and so we downgraded this evidence from moderate to low. More evidence is needed. |
| Maternal puerperal sepsis | RR 0.20 (0.05 ‐ 0.84) | 60 (one study**) | ⊕⊕⊕⊝ | **Defined as fever > 38° C and a positive high vaginal swab culture. |
| GRADE Working Group grades of evidence CI: confidence interval; CRP: C‐reactive protein; RR: risk ratio | ||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Persistant variable decelerations Show forest plot | 1 | 86 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.30, 0.91] |
| 2 Severe variable decelerations per hour in first stage Show forest plot | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | ‐1.2 [‐1.83, ‐0.57] |
| 3 Caesarean section Show forest plot | 2 | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only | |
| 3.1 Caesarean section overall | 2 | 147 | Risk Ratio (M‐H, Random, 95% CI) | 0.65 [0.25, 1.73] |
| 3.2 Caesarean section for fetal distress | 1 | 86 | Risk Ratio (M‐H, Random, 95% CI) | 0.43 [0.12, 1.55] |
| 4 Forceps/vacuum assisted delivery Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 Overall | 1 | 86 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.2 [0.58, 2.48] |
| 4.2 For fetal distress | 1 | 86 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.11 [0.01, 2.00] |
| 5 1 minute Apgar score < 4 Show forest plot | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.28 [0.03, 2.33] |
| 6 Umbilical arterial pH Show forest plot | 1 | 61 | Mean Difference (IV, Fixed, 95% CI) | 0.11 [0.08, 0.14] |
| 7 Umbilical pH ≤ 7.20 Show forest plot | 1 | 86 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.25 [0.06, 1.11] |
| 8 Neonatal morbidity Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 Overall | 1 | 86 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.19, 1.34] |
| 9 Neonatal death Show forest plot | 1 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.05, 5.77] |
| 10 Maternal puerperal sepsis Show forest plot | 2 | 147 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.36 [0.06, 2.18] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Fetal distress Show forest plot | 1 | 60 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.27 [0.08, 0.88] |
| 2 Gestational age at delivery (weeks) Show forest plot | 2 | 94 | Mean Difference (IV, Random, 95% CI) | ‐0.49 [‐2.63, 1.65] |
| 3 Neonatal morbidity Show forest plot | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 3.1 Neonatal sepsis/infection | 1 | 60 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.26 [0.11, 0.61] |
| 3.2 Pulmonary hypoplasia | 1 | 34 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.22 [0.06, 0.88] |
| 3.3 Abnormal neurological outcome | 1 | 34 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.25 [0.03, 2.01] |
| 4 Delivery within 7 days Show forest plot | 1 | 34 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.18 [0.05, 0.70] |
| 5 Time to delivery (days) Show forest plot | 1 | 60 | Mean Difference (IV, Fixed, 95% CI) | 0.57 [‐2.86, 4.00] |
| 6 Admission to neonatal intensive care unit Show forest plot | 1 | 34 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.90, 1.12] |
| 7 Neonatal death Show forest plot | 2 | 94 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.3 [0.14, 0.66] |
| 8 Birthweight (grams) Show forest plot | 2 | 94 | Mean Difference (IV, Random, 95% CI) | 15.65 [‐254.02, 285.32] |
| 9 Maternal puerperal sepsis Show forest plot | 1 | 60 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.2 [0.05, 0.84] |

