Fármacos catecolaminérgicos no antipsicóticos para la discinesia tardía inducida por antipsicóticos
Información
- DOI:
- https://doi.org/10.1002/14651858.CD000458.pub3Copiar DOI
- Base de datos:
-
- Cochrane Database of Systematic Reviews
- Versión publicada:
-
- 18 enero 2018see what's new
- Tipo:
-
- Intervention
- Etapa:
-
- Review
- Grupo Editorial Cochrane:
-
Grupo Cochrane de Esquizofrenia
- Copyright:
-
- Copyright © 2018 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Cifras del artículo
Altmetric:
Citado por:
Autores
Contributions of authors
Hany El‐Sayeh ‐ protocol updating, searching, trial selection, data extraction and assimilation (original version).
John Rathbone ‐ selected studies, data extraction, data assimilation (original version).
Karla Soares‐Weiser ‐ protocol writing, searching, trial selection, data extraction and assimilation (original version).
Hanna Bergman ‐ 2017 update: trial selection, data extraction and assimilation, 'Summary of findings' tables, report writing (2017 update).
Sources of support
Internal sources
-
CAPES ‐ Ministry of Education, Brazil.
-
Universidade Federal de Sao Paulo, Brazil.
-
Academic Unit of Psychiatry, Leeds., UK.
-
Enhance Reviews Ltd., UK.
Logistics support for Hanna Bergman for the 2016 update.
External sources
-
NIHR HTA Project Grant, reference number: 14/27/02, UK.
Salary support for Hanna Bergman.
Support for patient involvement consultation.
Support for traceable data database.
Declarations of interest
None known.
KSW is the Deputy Editor‐in‐Chief for Cochrane and Cochrane Innovations. When the NHIR HTA programme grant relevant to this review update was awarded, KSW was the Managing Director of Enhance Reviews Ltd.
HB worked for Enhance Reviews Ltd. during preparation of this review and was paid for her contribution to this review. Enhance Reviews Ltd. was a private company that performs systematic reviews of literature. HB works for Cochrane Response, an evidence consultancy that takes commissions from healthcare guideline developers and policy makers.
Acknowledgements
We would like to thank Clive Adams, Gill Rizzello and Tessa Grant for their advice and technical support. Thanks also to Ben Gray for writing the Plain Language Summary, and to Farhad Sokraneh for carrying out the 2015 trial search. We would like to thank João Paulo Lyra da Silva for his contribution to the previously published version of this review. We are also grateful to Dawn‐Marie Walker, Ruth Sayers, Megan Lees, and Vanessa Pinfold from McPin Foundation for organising and holding the public‐ and patient‐involvement consultation with TD service users that contributed to selecting outcomes for the 'Summary of findings' tables and to guiding future research. Finally, we wish to thank Rosie Asher and Antonio Grande for screening literature and helping with data extraction for the 2017 update, and Nicholas Henschke, Linda Levi and Loukia Spineli for assisting with updating the report.
Version history
| Published | Title | Stage | Authors | Version |
| 2018 Jan 18 | Non‐antipsychotic catecholaminergic drugs for antipsychotic‐induced tardive dyskinesia | Review | Hany G El‐Sayeh, John Rathbone, Karla Soares‐Weiser, Hanna Bergman | |
| 2006 Jan 25 | Non‐neuroleptic catecholaminergic drugs for neuroleptic‐induced tardive dyskinesia | Review | Hany George El‐Sayeh, Joao Paolo Lyra da Silva, John Rathbone, Karla Soares‐Weiser | |
| 1997 Jan 20 | Non‐neuroleptic catecholaminergic drugs for neuroleptic‐induced tardive dyskinesia | Protocol | Joao Paolo Lyra da Silva, Karla Soares‐Weiser, John McGrath | |
Differences between protocol and review
The protocol as published with this review has evolved over time. The revisions of protocol are in line with the development of RevMan and in keeping with Cochrane guidance. We think the revisions have greatly improved and enhanced this review. We do not think, however, that it has materially affected our conduct of the review or interpretation of the results.
In the 2017 review update, the biggest changes to affect the review methods were to:
-
broaden the inclusion criteria, and add the comparison 'Non‐antipsychotic catecholaminergic drug vs other drug';
-
change the title from 'Non‐neuroleptic catecholaminergic drugs for neuroleptic‐induced tardive dyskinesia' to 'Non‐antipsychotic catecholaminergic drugs for antipsychotic‐induced tardive dyskinesia';
-
update list of outcomes following consultation with consumers; and
-
add 'Summary of findings' tables.
Previous methods are reproduced in Appendix 1.
Keywords
MeSH
Medical Subject Headings (MeSH) Keywords
- Adrenergic Uptake Inhibitors [therapeutic use];
- Anti‐Dyskinesia Agents [*therapeutic use];
- Antipsychotic Agents [*adverse effects];
- Celiprolol [therapeutic use];
- Disease Progression;
- Dopamine Antagonists [therapeutic use];
- Dyskinesia, Drug‐Induced [*drug therapy];
- Haloperidol [therapeutic use];
- Methyldopa [therapeutic use];
- Randomized Controlled Trials as Topic;
- Reserpine [therapeutic use];
- Tetrabenazine [therapeutic use];
- Tiapamil Hydrochloride [therapeutic use];
Medical Subject Headings Check Words
Humans;
PICO

Message from one of the participants of the Public and patient involvement consultation of service user perspectives on tardive dyskinesia research.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
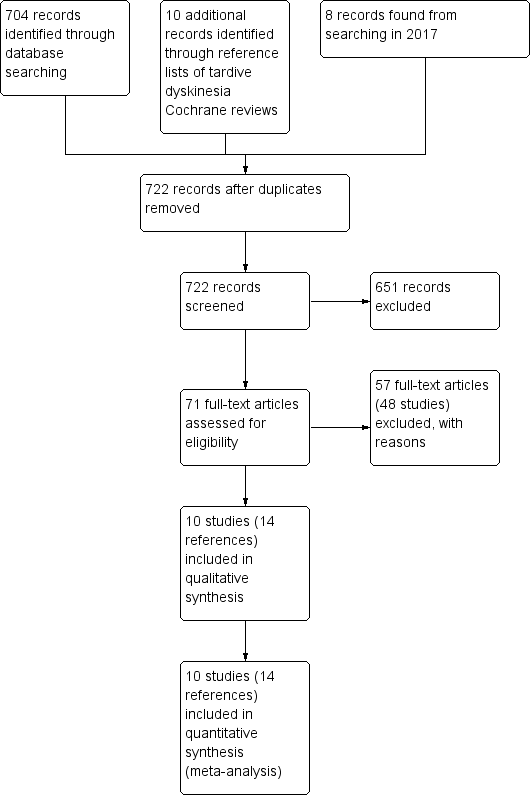
Study flow diagram for 2015 and 2017 searching
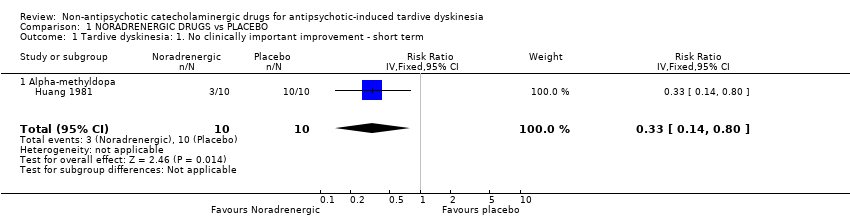
Comparison 1 NORADRENERGIC DRUGS vs PLACEBO, Outcome 1 Tardive dyskinesia: 1. No clinically important improvement ‐ short term.

Comparison 1 NORADRENERGIC DRUGS vs PLACEBO, Outcome 2 Tardive dyskinesia: 2. Not any improvement.

Comparison 1 NORADRENERGIC DRUGS vs PLACEBO, Outcome 3 Tardive dyskinesia: 3. Deterioration ‐ short term.
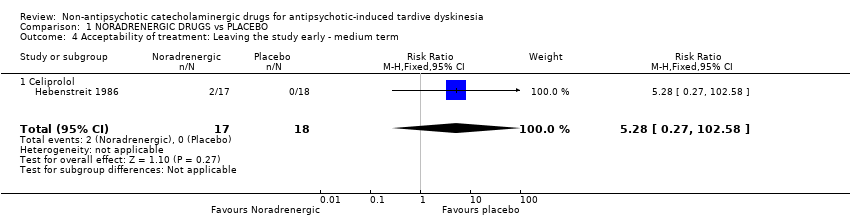
Comparison 1 NORADRENERGIC DRUGS vs PLACEBO, Outcome 4 Acceptability of treatment: Leaving the study early ‐ medium term.

Comparison 1 NORADRENERGIC DRUGS vs PLACEBO, Outcome 5 Quality of life: No improvement ‐ medium term.

Comparison 2 NORADRENERGIC DRUGS vs DOPAMINERGIC DRUGS, Outcome 1 Tardive dyskinesia: 1. No clinically important improvement ‐ short term.

Comparison 2 NORADRENERGIC DRUGS vs DOPAMINERGIC DRUGS, Outcome 2 Tardive dyskinesia: 2. Not any improvement ‐ short term.

Comparison 2 NORADRENERGIC DRUGS vs DOPAMINERGIC DRUGS, Outcome 3 Tardive dyskinesia: 3. Deterioration ‐ short term.

Comparison 3 DOPAMINERGIC DRUGS vs PLACEBO, Outcome 1 Tardive dyskinesia: 1. No clinically important improvement.

Comparison 3 DOPAMINERGIC DRUGS vs PLACEBO, Outcome 2 Tardive dyskinesia: 2. Not any improvement.
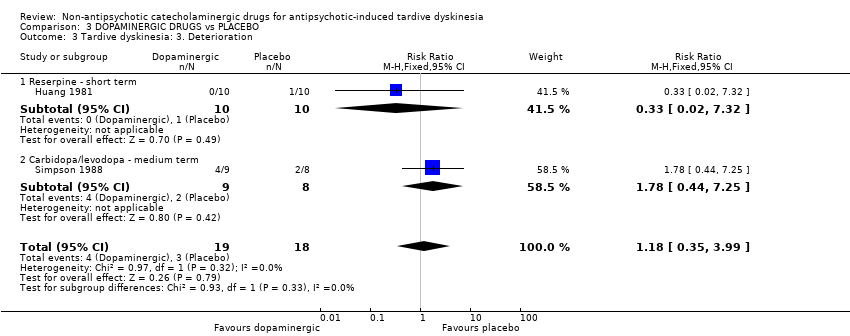
Comparison 3 DOPAMINERGIC DRUGS vs PLACEBO, Outcome 3 Tardive dyskinesia: 3. Deterioration.

Comparison 3 DOPAMINERGIC DRUGS vs PLACEBO, Outcome 4 Mental state: Deterioration ‐ medium term.
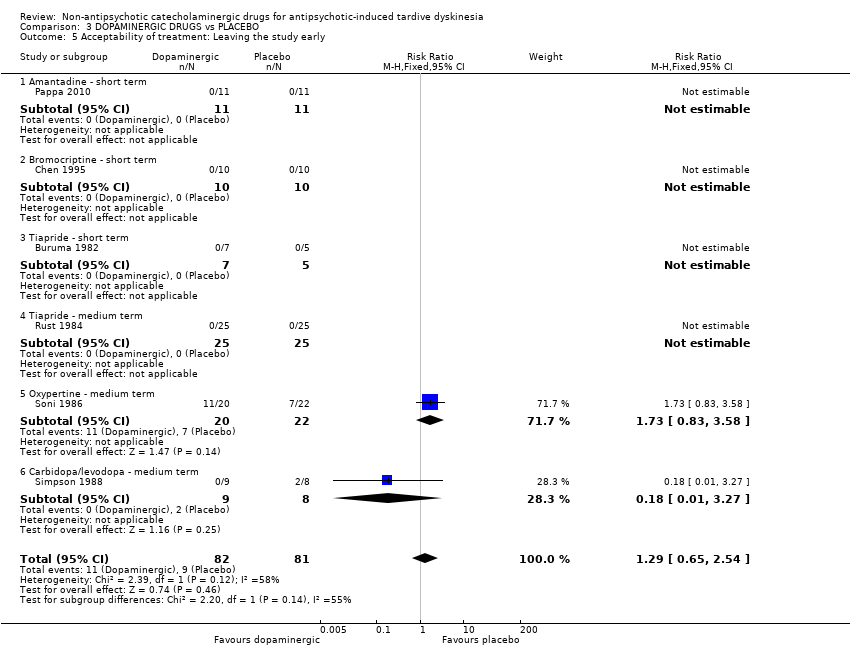
Comparison 3 DOPAMINERGIC DRUGS vs PLACEBO, Outcome 5 Acceptability of treatment: Leaving the study early.

Comparison 4 DOPAMINERGIC DRUGS vs OTHER DRUGS, Outcome 1 Tardive dyskinesia: 1. No clinically important improvement ‐ medium term.
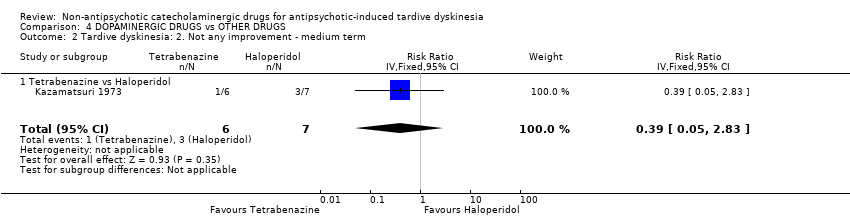
Comparison 4 DOPAMINERGIC DRUGS vs OTHER DRUGS, Outcome 2 Tardive dyskinesia: 2. Not any improvement ‐ medium term.

Comparison 4 DOPAMINERGIC DRUGS vs OTHER DRUGS, Outcome 3 Tardive dyskinesia: 3. Deterioration ‐ medium term.

Comparison 4 DOPAMINERGIC DRUGS vs OTHER DRUGS, Outcome 4 Acceptability of treatment: Leaving the study early ‐ medium term.
| Methods | Allocation: randomised, with sequence generation and concealment of allocation clearly described. |
| Participants | People with antipsychotic‐induced tardive dyskinesia.* |
| Interventions | 1. Non‐antipsychotic catecholaminergic compound. N = 150. |
| Outcomes | Tardive dyskinesia: any clinically important improvement in TD, any improvement, deterioration.*** |
| Notes | * This could be diagnosed by clinical decision. If funds were permitting all participants could be screened using operational criteria, otherwise a random sample should suffice. ** Size of study with sufficient power to highlight about a 10% difference between groups for primary outcome. |
| NORADRENERGIC DRUGS compared to PLACEBO for antipsychotic‐induced tardive dyskinesia | ||||||
| Patient or population: patients with antipsychotic‐induced tardive dyskinesia | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| PLACEBO | NORADRENERGIC DRUGS | |||||
| Tardive dyskinesia: No clinically important improvement follow‐up: 2 weeks | 1000 per 1000 | 330 per 1000 | RR 0.33 | 20 | ⊕⊕⊝⊝ | The included study evaluated alpha‐methyldopa. |
| Tardive dyskinesia: deterioration follow‐up: 2 weeks | 100 per 1000 | 33 per 1000 | RR 0.33 | 20 | ⊕⊝⊝⊝ | The included study evaluated alpha‐methyldopa. |
| Adverse events ‐ not reported | See comment | See comment | Not estimable | 0 | See comment | We found no studies rating this outcome. |
| Mental state ‐ not reported | See comment | See comment | Not estimable | 0 | See comment | We found no studies rating this outcome. |
| Acceptability of treatment: Leaving the study early follow‐up: 13 weeks | 0 per 1000 | 0 per 1000 | RR 5.28 | 35 | ⊕⊝⊝⊝ | The included study evaluated celiprolol. |
| No improvement in quality of life follow‐up: 13 weeks | 944 per 1000 | 822 per 1000 | RR 0.87 | 35 | ⊕⊝⊝⊝ | The included study evaluated celiprolol. |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded one step for risk of bias: unclear whether randomisation procedure and allocation concealment were carried out adequately, blinding of outcome assessors was not described. | ||||||
| NORADRENERGIC DRUGS compared to DOPAMINERGIC DRUGS for antipsychotic‐induced tardive dyskinesia | ||||||
| Patient or population: patients with antipsychotic‐induced tardive dyskinesia | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | № of participants | Quality of the evidence | Comments | |
| Risk with DOPAMINERGIC DRUGS | Risk with NORADRENERGIC DRUGS | |||||
| Tardive dyskinesia: No clinically important improvement follow‐up: 2 weeks | Study population | RR 0.60 | 20 | ⊕⊝⊝⊝ | ||
| 500 per 1,000 | 300 per 1,000 | |||||
| Tardive dyskinesia: Deterioration follow‐up: 2 weeks | Study population | not estimable | 20 | ⊕⊝⊝⊝ | Among the 20 participants no events were reported. | |
| 0 per 1,000 | 0 per 1,000 | |||||
| Adverse events ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| Mental state ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| Acceptability of treatment: Leaving the study early | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| Social confidence, social inclusion, social networks, or personalised quality of life ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded one step for risk of bias: unclear whether randomisation procedure and allocation concealment were carried out adequately. | ||||||
| DOPAMINERGIC DRUGS compared to PLACEBO for antipsychotic‐induced tardive dyskinesia | ||||||
| Patient or population: patients with antipsychotic‐induced tardive dyskinesia | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No of Participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| PLACEBO | DOPAMINERGIC DRUGS | |||||
| Tardive dyskinesia: No clinically important improvement follow‐up: 2 weeks | 1000 per 1000 | 520 per 1000 | RR 0.52 | 20 | ⊕⊕⊝⊝ | The included study evaluated reserpine. |
| Tardive dyskinesia: Deterioration follow‐up: 2‐6 weeks | 167 per 1000 | 197 per 1000 | RR 1.18 | 37 | ⊕⊝⊝⊝ | The included studies evaluated reserpine and carbidopa/levodopa. |
| Adverse events ‐ not reported | See comment | See comment | Not estimable | 0 | See comment | We found no studies rating this outcome. |
| General mental state: Deterioration follow‐up: 24 weeks | 45 per 1000 | 100 per 1000 | RR 2.2 | 42 | ⊕⊝⊝⊝ | The included study evaluated oxypertine. |
| Acceptability of treatment: Leaving the study early follow‐up: 2‐24 weeks | 111 per 1000 | 143 per 1000 | RR 1.29 | 163 | ⊕⊝⊝⊝ | Only two studies (59 participants) evaluating carbidopa/levodopa and oxypertine reported any events for this outcome. 4 studies evaluating amantadine, bromocriptine, and tiapride reported no events and consequently no estimates could be made for these 3 compounds. |
| Social confidence, social inclusion, social networks, or personalised quality of life ‐ not reported | See comment | See comment | Not estimable | 0 | See comment | This outcome was designated to be of importance, especially to patients. We found no studies rating this outcome. |
| *The basis for the assumed risk (e.g. the median control group risk across studies) is provided in footnotes. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded one step for risk of bias: unclear whether randomisation procedure and allocation concealment were carried out adequately, blinding of outcome assessors was not described. | ||||||
| DOPAMINERGIC DRUGS compared to OTHER DRUGS for antipsychotic‐induced tardive dyskinesia | ||||||
| Patient or population: patients with antipsychotic‐induced tardive dyskinesia | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | № of participants | Quality of the evidence | Comments | |
| Risk with OTHER DRUGS | Risk with DOPAMINERGIC DRUGS | |||||
| Tardive dyskinesia: No clinically important improvement follow‐up: 18 weeks | Study population | RR 0.93 | 13 | ⊕⊝⊝⊝ | ||
| 714 per 1000 | 664 per 1000 | |||||
| Tardive dyskinesia: Deterioration follow‐up: 18 weeks | Study population | RR 1.17 | 13 | ⊕⊝⊝⊝ | ||
| 143 per 1000 | 167 per 1000 | |||||
| Adverse events ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| Mental state ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| Acceptability of treatment: Leaving the study early follow‐up: 18 weeks | Study population | RR 0.23 | 13 | ⊕⊝⊝⊝ | ||
| 286 per 1000 | 66 per 1000 | |||||
| Social confidence, social inclusion, social networks, or personalised quality of life ‐ not reported | See comment | See comment | not estimable | 0 | See comment | We found no studies reporting on this outcome. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded one step for risk of bias: unclear whether randomisation procedure and allocation concealment were carried out adequately. | ||||||
| Interventions | Reference |
| Anticholinergic medication | |
| Benzodiazepines | |
| Calcium channel blockers | |
| Cholinergic medication | |
| Gamma‐aminobutyric acid agonists | |
| Miscellaneous treatments | |
| Neuroleptic reduction and/or cessation and neuroleptics | |
| Non‐neuroleptic catecholaminergic drugs | This review |
| Vitamin E |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Tardive dyskinesia: 1. No clinically important improvement ‐ short term Show forest plot | 1 | 20 | Risk Ratio (IV, Fixed, 95% CI) | 0.33 [0.14, 0.80] |
| 1.1 Alpha‐methyldopa | 1 | 20 | Risk Ratio (IV, Fixed, 95% CI) | 0.33 [0.14, 0.80] |
| 2 Tardive dyskinesia: 2. Not any improvement Show forest plot | 2 | 55 | Risk Ratio (IV, Fixed, 95% CI) | 0.91 [0.65, 1.27] |
| 2.1 Alpha‐methyldopa ‐ short term | 1 | 20 | Risk Ratio (IV, Fixed, 95% CI) | 0.33 [0.02, 7.32] |
| 2.2 Celiprolol ‐ medium term | 1 | 35 | Risk Ratio (IV, Fixed, 95% CI) | 0.92 [0.66, 1.28] |
| 3 Tardive dyskinesia: 3. Deterioration ‐ short term Show forest plot | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.02, 7.32] |
| 3.1 Alpha‐methyldopa | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.02, 7.32] |
| 4 Acceptability of treatment: Leaving the study early ‐ medium term Show forest plot | 1 | 35 | Risk Ratio (M‐H, Fixed, 95% CI) | 5.28 [0.27, 102.58] |
| 4.1 Celiprolol | 1 | 35 | Risk Ratio (M‐H, Fixed, 95% CI) | 5.28 [0.27, 102.58] |
| 5 Quality of life: No improvement ‐ medium term Show forest plot | 1 | 35 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.68, 1.12] |
| 5.1 Celiprolol | 1 | 35 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.87 [0.68, 1.12] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Tardive dyskinesia: 1. No clinically important improvement ‐ short term Show forest plot | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.6 [0.19, 1.86] |
| 1.1 Alpha‐methyldopa versus Reserpine | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.6 [0.19, 1.86] |
| 2 Tardive dyskinesia: 2. Not any improvement ‐ short term Show forest plot | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.1 Alpha‐methyldopa versus Reserpine | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Tardive dyskinesia: 3. Deterioration ‐ short term Show forest plot | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3.1 Alpha‐methyldopa versus Reserpine | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Tardive dyskinesia: 1. No clinically important improvement Show forest plot | 1 | 20 | Risk Ratio (IV, Fixed, 95% CI) | 0.52 [0.29, 0.96] |
| 1.1 Reserpine ‐ short term | 1 | 20 | Risk Ratio (IV, Fixed, 95% CI) | 0.52 [0.29, 0.96] |
| 2 Tardive dyskinesia: 2. Not any improvement Show forest plot | 3 | 57 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.60 [0.35, 1.03] |
| 2.1 Reserpine ‐ short term | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.02, 7.32] |
| 2.2 L‐DOPA ‐ short term | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.35, 1.27] |
| 2.3 Carbidopa/levodopa ‐ medium term | 1 | 17 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.26, 1.36] |
| 3 Tardive dyskinesia: 3. Deterioration Show forest plot | 2 | 37 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.18 [0.35, 3.99] |
| 3.1 Reserpine ‐ short term | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.02, 7.32] |
| 3.2 Carbidopa/levodopa ‐ medium term | 1 | 17 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.78 [0.44, 7.25] |
| 4 Mental state: Deterioration ‐ medium term Show forest plot | 1 | Risk Ratio (IV, Fixed, 95% CI) | Subtotals only | |
| 4.1 Oxypertine | 1 | 42 | Risk Ratio (IV, Fixed, 95% CI) | 2.2 [0.22, 22.45] |
| 5 Acceptability of treatment: Leaving the study early Show forest plot | 6 | 163 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.29 [0.65, 2.54] |
| 5.1 Amantadine ‐ short term | 1 | 22 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Bromocriptine ‐ short term | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.3 Tiapride ‐ short term | 1 | 12 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.4 Tiapride ‐ medium term | 1 | 50 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.5 Oxypertine ‐ medium term | 1 | 42 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.73 [0.83, 3.58] |
| 5.6 Carbidopa/levodopa ‐ medium term | 1 | 17 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.18 [0.01, 3.27] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Tardive dyskinesia: 1. No clinically important improvement ‐ medium term Show forest plot | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.93 [0.45, 1.95] |
| 1.1 Tetrabenazine vs Haloperidol | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.93 [0.45, 1.95] |
| 2 Tardive dyskinesia: 2. Not any improvement ‐ medium term Show forest plot | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.39 [0.05, 2.83] |
| 2.1 Tetrabenazine vs Haloperidol | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.39 [0.05, 2.83] |
| 3 Tardive dyskinesia: 3. Deterioration ‐ medium term Show forest plot | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 1.17 [0.09, 14.92] |
| 3.1 Tetrabenazine vs Haloperidol | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 1.17 [0.09, 14.92] |
| 4 Acceptability of treatment: Leaving the study early ‐ medium term Show forest plot | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.23 [0.01, 4.00] |
| 4.1 Tetrabenazine vs Haloperidol | 1 | 13 | Risk Ratio (IV, Fixed, 95% CI) | 0.23 [0.01, 4.00] |

