Cambios posturales periódicos durante la fototerapia en neonatos a término y prematuros con hiperbilirrubinemia
Resumen
Antecedentes
La fototerapia es la base del tratamiento de la hiperbilirrubinemia neonatal. El cambio postural periódico del neonato durante la fototerapia (de posición supina a posición prona o lateral) podría mejorar la eficiencia de la fototerapia al acelerar el acceso de la luz de la fototerapia a la bilirrubina depositada en diferentes partes de la piel y el tejido subcutáneo.
Objetivos
Evaluar los efectos del cambio postural periódico durante la fototerapia, en comparación con ningún cambio postural prescrito, sobre el nivel total de bilirrubina sérica y la duración del tratamiento en neonatos con hiperbilirrubinemia no conjugada durante los primeros 28 días de vida.
Los objetivos secundarios de la revisión incluyeron la evaluación de la eficacia del cambio postural periódico sobre la necesidad o el número de exanguinotransfusiones, la incidencia de daño neurológico inducido por la bilirrubina (DNIB), los efectos secundarios de la fototerapia y el síndrome de muerte súbita del lactante (SMSL).
Métodos de búsqueda
Se utilizó la estrategia de búsqueda estándar del Grupo Cochrane de Neonatología (Cochrane Neonatal) para realizar búsquedas exhaustivas en el Registro Cochrane central de ensayos controlados (Cochrane Central Register of Controlled Trials, CENTRAL; 2021, número 3) en la Biblioteca Cochrane y en Ovid MEDLINE y Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Daily and Versions el 5 de marzo de 2021. También se realizaron búsquedas en bases de datos de ensayos clínicos y en las listas de referencias de los estudios incluidos y de revisiones pertinentes para obtener ensayos controlados aleatorizados (ECA) y cuasialeatorizados.
Criterios de selección
Los ECA y los ensayos controlados cuasialeatorizados se incluyeron si reclutaron neonatos (a término y prematuros) de cualquier sexo con hiperbilirrubinemia no conjugada que requería fototerapia y compararon el cambio postural periódico del neonato durante la fototerapia con ningún cambio postural prescrito.
Obtención y análisis de los datos
Dos autores de la revisión de forma independiente evaluaron la calidad de los ensayos, extrajeron los datos y consultaron con un tercer autor de la revisión en caso de desacuerdos. Se utilizaron los procedimientos metodológicos estándar de Cochrane, incluida la evaluación del riesgo de sesgo de los estudios incluidos. Se utilizó el método GRADE para evaluar la certeza de la evidencia. Los desenlaces principales fueron la duración de la fototerapia y la tasa de descenso de la bilirrubina sérica a las 24 horas. Los desenlaces secundarios incluyeron la necesidad de exanguinotransfusión, el número de exanguinotransfusiones, la incidencia de DNIB y el SMSL.
Resultados principales
En la revisión se incluyeron cinco estudios (343 neonatos) con un alto riesgo general de sesgo. La posición corporal durante la fototerapia se cambió cada dos horas o cada dos horas y media en dos estudios, y cada tres horas en un estudio. Tres de los cinco estudios incluyeron neonatos sanos a término, mientras que los otros dos estudios también incluyeron neonatos prematuros (≥ 33 semanas de gestación); sin embargo, no se dispuso de datos separados sobre los desenlaces de la revisión en neonatos prematuros.
El cambio postural periódico podría dar lugar a poca o ninguna diferencia en la duración de la fototerapia (diferencia de medias [DM] 1,71 horas; intervalo de confianza [IC] del 95%: ‐3,17 a 6,59 horas; I² = 58%; cuatro estudios, 231 participantes; evidencia de certeza baja). Sólo un estudio informó sobre la tasa de descenso de la bilirrubina total sérica a las 24 horas de iniciar la fototerapia. El cambio postural periódico podría dar lugar a poca o ninguna diferencia en la tasa de descenso de la bilirrubina total sérica a las 24 horas (DM 0,02 mg/dl/h; IC del 95%: ‐0,02 a 0,06 mg/dl/h; un estudio, 100 participantes; evidencia de certeza baja). La certeza de la evidencia se disminuyó a muy baja debido al riesgo de sesgo y la imprecisión. Ninguno de los estudios incluidos informó sobre la necesidad o el número de exanguinotransfusiones, la incidencia de DNIB ni el SMSL. La falta de datos separados impidió realizar un análisis de subgrupos.
Conclusiones de los autores
La evidencia disponible no es suficientes para determinar los efectos del cambio postural periódico en comparación con ningún cambio postural prescrito en la fototerapia. Hay evidencia de certeza baja de que podría haber poca o ninguna diferencia en la duración de la fototerapia y la tasa de descenso de la bilirrubina a las 24 horas de iniciada la fototerapia entre el cambio postural periódico y ningún cambio postural prescrito durante la fototerapia en neonatos a término y prematuros. Ninguno de los estudios incluidos informó sobre el efecto del cambio postural sobre la necesidad o el número de exanguinotransfusiones, la incidencia de DNIB ni el SMSL. Un estudio está pendiente de clasificación y no se pudo incluir en la revisión. Se necesitan más estudios para evaluar el efecto del cambio postural periódico en la fototerapia, especialmente en neonatos con hiperbilirrubinemia hemolítica y en neonatos muy prematuros. Los resultados de esta revisión sistemática se aplican principalmente a los neonatos prematuros tardíos o a término que reciben fototerapia por hiperbilirrubinemia no hemolítica.
PICO
Resumen en términos sencillos
Cambios posturales periódicos durante la fototerapia en neonatos a término y prematuros con hiperbilirrubinemia
Pregunta de la revisión
¿El cambio de posición mejora los resultados de la fototerapia en los recién nacidos a término y prematuros con ictericia?
Mensajes clave
En esta revisión sistemática se evaluaron los estudios que compararon el cambio de posición programado y ningún cambio de posición programado de los recién nacidos a término y prematuros que recibieron fototerapia para la ictericia. Estas dos estrategias de cambio de posición dieron lugar a poca o ninguna diferencia en la duración de la fototerapia o en la velocidad de descenso de los niveles de bilirrubina. La bilirrubina es la sustancia que provoca la ictericia, es decir, el color amarillo de la piel y de la parte blanca de los ojos.
Ninguno de los estudios incluidos en la revisión informó del efecto que tiene cambiar la posición de los recién nacidos sobre los efectos no deseados, incluido el síndrome de muerte súbita del lactante.
Los estudios futuros deberían investigar el cambio de posición programado de los recién nacidos durante la fototerapia, e incluir a recién nacidos muy prematuros y a recién nacidos que presenten todos los tipos de ictericia.
¿Qué es la ictericia?
La ictericia (también llamada hiperbilirrubinemia) es una enfermedad frecuente en los recién nacidos, que hace que la piel y la parte blanca de los ojos se vuelvan amarillos como resultado de un exceso de bilirrubina en la sangre. La bilirrubina es una sustancia amarilla que se produce cuando los glóbulos rojos se descomponen. Como el hígado de los recién nacidos no puede eliminar la bilirrubina de la sangre de forma eficaz, los niveles se acumulan. En algunos casos, los recién nacidos alcanzan niveles muy altos de bilirrubina en sangre, lo que puede causar daños cerebrales. Una vez que los recién nacidos tienen unas dos semanas de vida, su hígado puede procesar la bilirrubina y la ictericia mejora por sí sola.
¿Cómo se trata la ictericia?
El tratamiento más común para la ictericia es la terapia con luz (fototerapia). Los recién nacidos se colocan bajo una luz especial, con los ojos tapados, llevando sólo un pañal, para que la mayor cantidad de piel posible quede expuesta a la luz. La fototerapia descompone la bilirrubina, que puede ser eliminada por el organismo. Algunos recién nacidos pueden sufrir sarpullidos o diarrea con la fototerapia, pero no suele causar efectos indeseados. Normalmente se necesitan unas 48 horas de fototerapia para reducir la bilirrubina a un nivel seguro en la mayoría de los recién nacidos.
¿Qué se quería averiguar?
Al girar a los recién nacidos sobre su espalda y luego sobre el costado durante la fototerapia, se exponen diferentes zonas de la piel a la luz y podrían reducir los niveles de bilirrubina más rápidamente. Se quería averiguar si la fototerapia es más eficaz si se voltea a los recién nacidos a horas determinadas, en comparación con mantenerlos en una sola posición.
Interesaba determinar especialmente los efectos en los recién nacidos a término (hasta tres semanas antes de la fecha prevista de parto) y en los recién nacidos prematuros, durante los primeros 28 días de vida. Se quería saber si el cambio de posición de los recién nacidos reduce el tiempo necesario de fototerapia, cambia la velocidad de descenso de la bilirrubina o si causa algún efecto no deseado, especialmente el síndrome de muerte súbita del lactante (SMSL).
¿Qué se hizo?
Se buscaron estudios que investigaran cambiar la posición del cuerpo en comparación con no hacerlo durante la fototerapia en recién nacidos con ictericia. Los cambios de posición del cuerpo de los recién nacidos y los tiempos entre los cambios debían seguir un horario establecido. Los recién nacidos debían presentar ictericia, nacer a término o ser prematuros, y podían ser de sexo masculino o femenino.
¿Qué se encontró?
Se encontraron cinco estudios con un total de 343 recién nacidos. Tres estudios incluyeron a recién nacidos sanos y a término, mientras que los otros dos estudios incluyeron a recién nacidos a término y prematuros. Todos los estudios compararon cambios programados de la posición del cuerpo de los recién nacidos con cambiarla durante la fototerapia. Los recién nacidos se podían colocar boca arriba, boca abajo o de lado, y su posición se cambiaba según el plan del estudio. Los cambios de posición se basaron en el tiempo (cada 30 minutos a seis horas); la manipulación del recién nacido (p. ej., después de cada sesión de lactancia); o el cambio en cada turno de enfermería (p. ej., una vez por turno).
Resultados principales
Cambiar la posición del cuerpo de los recién nacidos durante la fototerapia, en comparación con no cambiarla, podría suponer una diferencia mínima o nula en la duración de la fototerapia (cuatro estudios, 231 recién nacidos) y en la velocidad de descenso de los niveles de bilirrubina a las 24 horas de comenzar la fototerapia (un estudio, 100 recién nacidos). No se sabe si el cambio de posición causa efectos no deseados, ya que ninguno de los estudios incluidos informó sobre ello.
¿Cuáles son las limitaciones de la evidencia?
La confianza en la evidencia es limitada porque se encontraron pocos estudios, y los que se encontraron no utilizaron los mejores métodos para analizar y comunicar sus resultados. Ninguno de los estudios informó sobre los efectos no deseados.
¿Cuál es el grado de actualización de esta evidencia?
La evidencia está actualizada hasta el 5 de marzo de 2021.
Authors' conclusions
Summary of findings
| Turning position versus supine under phototherapy in term and preterm neonates with hyperbilirubinaemia | ||||||
| Patient or population: term and preterm neonates with hyperbilirubinaemia | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | No. of participants | Certainty of the evidence | Comments | |
|---|---|---|---|---|---|---|
| Risk with turning position under phototherapy | Risk with supine position under phototherapy | |||||
| Duration of phototherapy (hours) | The mean duration of phototherapy ranged from 24.8 to 52.8 hours. | The mean duration of phototherapy ranged from 25.5 to 53.3 hours. | Mean duration of phototherapy was on average 1.71 hours longer in the turning group (95% CI −3.17 to 6.59). | 231 | ⊕⊕⊝⊝ | Periodic change in body position may lead to little or no difference in the duration of phototherapy. |
| Rate of fall of serum total bilirubin at 24 hours of starting phototherapy (mg/dL/h)b | The mean rate of fall of serum total bilirubin at 24 hours of starting phototherapy was 0.22 mg/dL/h. | The mean rate of fall of serum total bilirubin at 24 hours of starting phototherapy was 0.2 mg/dL/h. | MD in the rate of fall of serum total bilirubin was an average of 0.02 mg/dL/h higher in the turning group | 100 | ⊕⊕⊝⊝ Lowc | Periodic change in body position may lead to little or no effect on the rate of fall of serum total bilirubin at 24 hours of starting phototherapy. |
| Need for exchange transfusion | None of the studies reported this outcome. | |||||
| Bilirubin‐induced neurological damage (BIND) | None of the studies reported this outcome. | |||||
| Sudden infant death syndrome (SIDS) | None of the studies reported this outcome. | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| aDowngraded by two levels because of statistical heterogeneity and unclear risk of selection bias. (One study has attrition bias. Three studies have unclear reporting. Blinding of outcome assessors was not possible in any of the studies.) | ||||||
Background
Description of the condition
'Hyperbilirubinaemia' is a term used to describe elevated levels of bilirubin in the blood. Nearly 97% of full‐term and preterm babies demonstrate biochemical hyperbilirubinaemia (defined as serum bilirubin level > 1 mg/dL), and more than two‐thirds appear clinically icteric (serum bilirubin > 5 mg/dL) (Maisels 1986). In the majority of cases, hyperbilirubinaemia is physiological due to increased breakdown of red blood cells, decreased uptake and metabolism of bilirubin in the liver, and increased enterohepatic circulation (Wong 2019). However, the presence of conditions like blood group incompatibility, glucose‐6‐phosphate dehydrogenase enzyme deficiency, the presence of extravasated blood, and suboptimal feeding can cause an excessive rise of bilirubin (Wells 2013). Bilirubin is a potential neurotoxin, and severe hyperbilirubinaemia can result in neurological damage (Bhutani 2013; Cashore 1990).
The acute phase of bilirubin‐induced neurological damage (BIND) manifests clinically with poor feeding, lethargy, a high‐pitched cry, hypertonia or hypotonia, opisthotonos, and seizures. The chronic manifestations include athetoid cerebral palsy, motor delay, gaze palsy, dental dysplasia, mental retardation, and sensorineural hearing loss. Prematurity, lower postnatal age, ongoing haemolysis, low albumin levels, and other comorbidities like asphyxia and sepsis may increase the risk of BIND. Threshold bilirubin levels for initiating treatment for hyperbilirubinaemia are therefore based on gestation at birth, postnatal age, and the presence of risk factors (AAP 2004).
Description of the intervention
Phototherapy is the standard treatment of neonatal hyperbilirubinaemia (Maisels 2008; Polin 1990). Phototherapy is provided by placing a light source over the baby bassinet. Bilirubin present in the skin, subcutaneous tissue, and peripheral capillaries absorbs the light energy emitted by the phototherapy unit and as a result of different photochemical reactions, is converted into more easily excreted photo‐isomers. If phototherapy fails to reduce bilirubin concentration below the neurotoxic levels, exchange transfusion is performed. However, exchange transfusion is associated with a risk of death (nearly 2%) or severe complications (12%) (Jackson 1997).
Various strategies employed to improve the efficacy of phototherapy include using a high‐intensity light source, decreasing the distance between the baby and the light source, using double‐surface phototherapy (application of another panel angulated to the first panel), and increasing the surface area of skin exposed to phototherapy (Pratesi 1989; Vreman 2004). The posture of the baby under phototherapy is usually governed by nursing preferences and the hospital policy to decrease the risk of sudden infant death syndrome (SIDS) (Jeffery 1999). As a result, neonates are most likely to be nursed in a supine posture under phototherapy. Parts of the skin under phototherapy are in a state of continuous equilibrium with the actual 'bilirubin pool' of the body such that the bilirubin photo‐isomers (formed after phototherapy) are constantly being removed from the skin and added to the circulation pool, and fresh unconjugated bilirubin circulating in the blood pool is being added to the skin (both along their respective concentration gradients). It can be hypothesised that periodically changing the body position from supine to prone or lateral positions may improve the efficiency of phototherapy by hastening the access of phototherapy light to bilirubin deposited in various parts of the skin and subcutaneous tissue.
How the intervention might work
The principal site of action of phototherapy is the outermost 2 mm of the skin (Vogl 1974). First, bilirubin in the skin is converted to its photo‐isomers, a process that occurs in nanoseconds (Cremer 1958). The photo‐isomer conversion includes configurational isomerisation, structural isomerisation, and photo‐oxidation. Configurational isomerisation is a very rapid process that changes bilirubin isomers to water‐soluble isomers; however, this isomerisation is not significantly influenced by light intensity (Dennery 2001; Maisels 2008). On the other hand, structural isomerisation, which consists of intramolecular cyclisation resulting in the formation of lumirubin, is enhanced by increasing the intensity of light. The second step is the migration of these photo‐isomers into the circulation and simultaneously, the reverse migration of non‐isomerised serum bilirubin into the skin (Cremer 1958; Maisels 2001). This second step is considered rate‐limiting, and the time required is estimated at between one and three hours (Lau 1984; Vogl 1974). Assuming that this migration occurs only through skin‐blood‐skin bilirubin transfer, and there is no lateral diffusion of bilirubin from unexposed skin to the adjacent exposed skin, phototherapy becomes less effective once the exposed skin is blanched. Hence, turning the infant over from the blanched side to the bilirubin‐loaded side (the side not receiving phototherapy) would seem to be a logical therapeutic manoeuvre for increasing the rate of bilirubin degradation and thereby improving the efficacy of phototherapy (Lau 1984; Vogl 1978).
Why it is important to do this review
The American Academy of Pediatrics (AAP) Subcommittee on Hyperbilirubinemia guidelines, AAP 2004, and the National Institute for Health and Care Excellence (NICE) guidelines on neonatal jaundice, NICE 2016, do not provide any recommendation for or against changing the infant's position under phototherapy (AAP 2004; Rennie 2010). The NICE guidelines recommend supine positioning, and the AAP guidelines do not recommend any specific position for infants. In view of conflicting evidence from studies and uncertain clinical practice regarding the optimal position of infants under phototherapy, the search for the optimal position during phototherapy remains an important research priority. The authors of a systematic review on the effect of turning versus supine position for neonates under phototherapy suggested that keeping the jaundiced newborns in the supine position throughout phototherapy is as effective as turning them (Lee Wan Fei 2015). However, this systematic review excluded studies not available in the English language and studies with position changes more frequent than two‐ to three‐hourly. Lee Wan Fei 2015 did not consider patient‐important outcomes such as BIND, need or the number of exchange transfusions, kernicterus, or SIDS. Any recommendation on change in body position during phototherapy also needs to consider a possible association with the risk of SIDS (Hunt 2003; Mitchell 1999).
Objectives
To evaluate the effects of periodic change of body position during phototherapy as compared to no prescribed change in body position, on serum total bilirubin level and duration of treatment in neonates with unconjugated hyperbilirubinaemia during the first 28 days of life.
Secondary objectives of the review included evaluation of the efficacy of periodic change of body position on the need for or number of exchange transfusions, incidence of bilirubin‐induced neurological damage (BIND), side effects of phototherapy, and sudden infant death syndrome (SIDS).
Methods
Criteria for considering studies for this review
Types of studies
We included randomised controlled trials (RCTs) and quasi‐RCTs comparing periodic change in body position of the infant under phototherapy to no prescribed change in body position. We did not apply any restrictions on language or sex. Trials reported in abstract form were eligible for inclusion if methods were reported that allowed assessment of eligibility for inclusion and risk of bias.
We did not plan to include cross‐over trials, as this is not an appropriate study design to answer the review question. We also planned to exclude cluster‐RCTs, as this study design, although theoretically feasible, is unlikely to be used for the review question.
Types of participants
We included studies enrolling neonates (term (≥ 37 weeks' gestation) and preterm (< 37 weeks' gestation)) of either gender with unconjugated hyperbilirubinaemia (irrespective of aetiology and defined as hyperbilirubinaemia with direct‐reacting component bilirubin level > 1 mg/dL or > 20% of the total bilirubin (Feldman 2014)) within the first 28 days of life.
Types of interventions
We included studies comparing the periodic change in body position of the infant under phototherapy with no prescribed change in body position. The infant’s position may include supine, prone, or lateral body posture with the intervention prescribing a periodic change in position. Periodicity for change in position may be based on time (every 30 minutes to 6 hours), handling of the baby (e.g. after each breastfeeding session), or nursing shift (e.g. once during a shift).
Types of outcome measures
We included the following outcomes in this review.
Primary outcomes
-
Duration of phototherapy (hours)
-
Rate of fall of serum total bilirubin (mg/dL/h) at 24 hours
Secondary outcomes
-
Need for exchange transfusion (proportion)
-
Number of exchange transfusions (mean number)
-
Incidence of bilirubin‐induced neurological damage (BIND) (proportion). BIND or subtle encephalopathy was defined as neurological, cognitive, learning, movement disorders, isolated hearing loss or auditory dysfunction in the presence of hyperbilirubinaemia (Bergman 1985; Hyman 1969; Johnson 1974; Rubin 1979; Scheldt 1977)
-
Phototherapy side effects, including skin rash
-
Kernicterus (Dennery 2001; Gkoltsiou 2008; Shapiro 2005)
-
Sudden infant death syndrome (SIDS) (proportion): SIDS was defined as “the sudden death of an infant under one year of age, which remains unexplained after a thorough case investigation, including the performance of a complete autopsy, examination of the death scene, and review of the clinical history” (Willinger 1991).
Search methods for identification of studies
Electronic searches
We conducted a comprehensive search without date, language, or publication type limits in March 2021. We searched the following databases: Cochrane Central Register of Controlled Trials (CENTRAL; 2021, Issue 3) in the Cochrane Library and Ovid MEDLINE(R) and Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Daily and Versions(R) (1 January 2016 to 5 March 2021). The strategy combined terms for hyperbilirubinaemia and phototherapy combined with standard neonatal terms and a filter for randomised controlled trials. Search strategies are provided in Appendix 1.
We searched clinical trial registries for ongoing or recently completed trials on 5 March 2021. We searched the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) (www.who.int/ictrp/search/en/) and the US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (clinicaltrials.gov), via Cochrane CENTRAL. We also searched the ISRCTN registry (www.isrctn.com/), for any unique trials not found through the CENTRAL search.
Our previous search details (up to May 2016) are listed in Appendix 2.
Searching other resources
We cross‐referenced relevant literature including identified trials and existing review articles. We searched the following additional resources:
-
reference lists of articles retrieved from the sources cited under Electronic searches and from review articles;
-
personal communication with primary authors of studies to retrieve unpublished data related to published articles.
Data collection and analysis
We used the standard methods of Cochrane and Cochrane Neonatal.
Selection of studies
Two review authors (AT and DC) independently assessed the studies identified by the search for inclusion in the review using the Covidence interface (Covidence). Any disagreements were resolved through mutual discussion.
Data extraction and management
Two review authors (AT and DC) independently extracted data using a pre‐tested data extraction form. Any disagreements were resolved through mutual discussion or by consulting a third review author (AD) if necessary.
Assessment of risk of bias in included studies
Two review authors (AT and DC) independently assessed the risk of bias (low, high, or unclear) of all included trials using the Cochrane risk of bias tool (Higgins 2011), for the following domains:
-
sequence generation (selection bias);
-
allocation concealment (selection bias);
-
blinding of participants and personnel (performance bias);
-
blinding of outcome assessment (detection bias);
-
incomplete outcome data (attrition bias);
-
selective reporting (reporting bias);
-
any other bias.
Any disagreements were resolved through mutual discussion or by consulting a third review author if necessary. See Appendix 3 for a more detailed description of risk of bias for each domain.
Measures of treatment effect
We analysed continuous data using mean difference (MD). We calculated the risk ratio (RR), risk difference (RD), the number needed to treat for an additional beneficial outcome (NNTB), and the number needed to treat for an additional harmful outcome (NNTH) with 95% confidence intervals (CI) for dichotomous data.
Unit of analysis issues
We compared periodic change in body position of the infant under phototherapy with no prescribed change in body position. The infant’s position could include supine, prone, or lateral body posture, with the intervention prescribing a periodic change in position. Periodicity for change in position may be based on time (every 30 minutes to 6 hours), handling of the baby (e.g. after each breastfeeding session), or nursing shift (e.g. once during a shift).
In the current review, the interval between changes in body position varied from two to three hours. All of the included studies compared 'change' versus 'no prescribed change' in body position, not different protocols for body position. If in future updates of the review we find multi‐arm studies comparing different protocols for change in body position versus no prescribed change, we will combine the intervention groups to create a single pair‐wise comparison of intervention (any prescribed change) with the control group (no prescribed change).
Dealing with missing data
We contacted the authors of studies awaiting classification for any published or presented version of the study results (IRCT201111063250N4); however, the results of the study have not been published. The authors of the five studies included in the review were not contacted for any missing data.
Assessment of heterogeneity
We examined heterogeneity between trials by first assessing differences in trial methodologies and clinical heterogeneity. We used the following cut‐offs and labels for the results of the I² test:
-
< 25%: no heterogeneity;
-
25% to 49%: low heterogeneity;
-
50% to 74%: moderate heterogeneity;
-
> 74%: high heterogeneity.
We planned to inspect forest plots and quantify the impact of heterogeneity using the I²statistic. If we found heterogeneity, we would explore the possible causes of statistical heterogeneity using prespecified subgroup analysis (e.g. term versus preterm neonates and haemolytic versus non‐haemolytic jaundice). We used a fixed‐effect model for meta‐analyses.
Assessment of reporting biases
We planned to use funnel plots to assess for publication bias if there were a sufficient number (> 10) of trials reporting the same outcome. However, we included only five trials in the review, which precluded an assessment of publication bias.
Data synthesis
We tabulated studies that met the inclusion criteria to enable the comparison of trial characteristics and individual components of the quality assessment. We tabulated the bibliographic details of trials excluded from the review with documentation of the specific reasons for exclusion. We reviewed the summary tables of included trials to identify clinical heterogeneity amongst trials. We implemented a meta‐analysis with a random‐effects model using Review Manager 5 software (Review Manager 2014).
Subgroup analysis and investigation of heterogeneity
We planned subgroup analysis to consider:
-
term (≥ 37 weeks') versus preterm (< 37 weeks') neonates;
-
haemolytic versus non‐haemolytic hyperbilirubinaemia.
In addition, we planned to carry out subgroup analysis if studies reported different regimens of change in position (e.g. frequency of change in position or change in position under a single‐surface versus double‐surface phototherapy). However, subgroup analysis was not possible, as separate data for the subgroups were not reported in any of the included studies.
Sensitivity analysis
We planned to perform sensitivity analyses to test the robustness of our decisions if a sufficient number of trials were found. We planned to perform a sensitivity analysis if the findings were affected by including only studies of adequate methodology (low risk of bias), defined as adequate randomisation and allocation concealment, blinding of intervention and outcome measurement, and less than 10% loss to follow‐up. However, we did not perform sensitivity analysis, as the five studies included in the review had similar methodological rigour.
Summary of findings and assessment of the certainty of the evidence
We used the GRADE approach as outlined in the GRADE Handbook to assess the certainty of the evidence for the following outcomes (Schünemann 2013):
-
duration of phototherapy;
-
per cent fall in bilirubin at 24 hours of starting phototherapy;
-
rate of fall of serum bilirubin at 24 hours of starting phototherapy;
-
serum total bilirubin at 24 hours of starting phototherapy.
Two review authors (AT and DC) independently assessed the certainty of evidence for each of the listed outcomes above. We considered evidence from RCTs as high certainty, downgrading one level for serious (or two levels for very serious) limitations based upon the following: design (risk of bias), consistency across studies, directness of the evidence, precision of results, and presence of publication bias. We used GRADEpro GDT software to create summary of findings Table 1 to report the certainty of the evidence (GRADEpro GDT). The GRADE approach results in an assessment of the certainty of a body of evidence as one of four grades.
-
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
-
Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
-
Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect.
-
Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect.
Results
Description of studies
See Characteristics of included studies; Characteristics of excluded studies; Characteristics of studies awaiting classification.
Results of the search
Our literature search in March 2021 identified a total of 1472 records. After the removal of duplicates, we screened 1228 records, of which 1216 records were excluded after title and abstract screening. We screened the full texts of 12 studies, excluding six studies for a variety of reasons. One record from a trial registry has not been published and was categorised as awaiting classification (IRCT201111063250N4). We included five studies in the review.
See Figure 1.
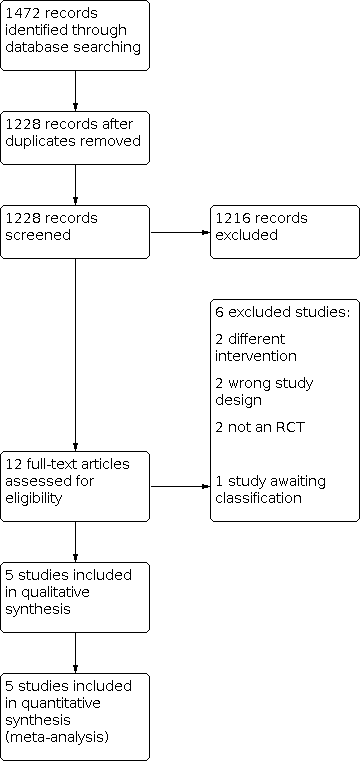
Study flow diagram.
Included studies
We included five studies (343 neonates) in the review.
Bhethanabhotla 2013 enrolled neonates born at 35 to 42 weeks of gestation, with age > 24 hours and < 14 days and hyperbilirubinaemia requiring phototherapy. Enrolled neonates were randomised to either no change in position (n = 54) or alternating supine and prone position change every two hours (n = 46) whilst under phototherapy. Both groups received single‐surface phototherapy with four blue and two white compact fluorescent tube lights. The light source was kept at a 25‐centimetre distance from the neonate. Neonates in both groups received exclusive breastfeeding. Baseline variables like gestation, birth weight, and bilirubin at the onset of phototherapy were comparable in the two groups. Reported outcomes included the duration of phototherapy, absolute levels of serum total bilirubin (STB), and rate of fall of STB at 24 hours of starting phototherapy.
Chen 2002 enrolled clinically well, term infants with non‐haemolytic hyperbilirubinaemia (bilirubin level greater than 15 mg/dL). Criteria for the diagnosis of non‐haemolytic hyperbilirubinaemia included: absence of blood group isoimmunisation, a negative Coombs' test, haemoglobin concentration greater than 14 g/dL, and a normal glucose‐6‐phosphate dehydrogenase status. Neonates were randomised to a "fixed" supine position group (n = 24) or the "position‐changing" turning group (n = 27). In the supine group, neonates were kept in supine posture whilst under phototherapy. In the turning group, the position of neonate under phototherapy was rotated from a supine to a prone position or vice versa every two hours. Both groups received single‐surface phototherapy with six white compact fluorescent tube lights. The light source was kept at a 35‐centimetre distance from the neonate. Phototherapy was interrupted for feeding. Baseline variables like birth weight, gestational age, gender, feeding type, age at phototherapy, haemoglobin at the start of phototherapy, and measured spectral irradiance were comparable in the two groups. Outcomes reported by the study included the duration of phototherapy, per cent fall in STB at 24 hours and 48 hours of phototherapy, and the rate of fall of STB (time not specified).
Donneborg 2010 enrolled healthy neonates with non‐haemolytic hyperbilirubinaemia, gestation ≥ 33 weeks, fulfilling the indications for phototherapy, postnatal age > 24 hours, not having received phototherapy for the last 48 hours, and who were able to be treated in a cradle. Enrolled neonates were randomised to supine (n = 53) or alternating position (turning, n = 59) groups. In the supine group, neonates were kept in supine posture whilst under phototherapy. In the turning group, the position was changed every third hour from supine to prone and vice versa. Both groups received single‐surface phototherapy with a light‐emitting device (LED) emitting blue light with an emission peak at 470 nm and a bandwidth of 455 to 485 nm. The light source was kept at a 20‐centimetre distance from the neonate. Neonates in both groups received feeding for 30 minutes every third hour. The outcomes reported included absolute levels of STB and its per cent fall at 24 hours and 48 hours of starting phototherapy.
Mohammadzadeh 2004 enrolled healthy, exclusively breastfed neonates more than 48 hours of postnatal age, with birth weight > 2500 g, born at 38 to 41 weeks of gestation after an uncomplicated pregnancy, and who had indirect hyperbilirubinaemia with STB ≥ 15 mg/dL at 49 to 72 hours of age or ≥ 17 mg/dL at ≥ 72 hours of age. Neonates with haemolytic disease, infection, congenital anomaly, closed haemorrhage (cephalohaematoma), and metabolic diseases were excluded. Enrolled neonates were randomised to supine (n = 25) or turning (n = 25) groups. In the supine group, neonates were kept in supine posture whilst under phototherapy. In the turning group, the position of the neonate under phototherapy was changed from supine to prone every 150 minutes, followed by a break of 30 minutes for feeding and routine nursing care. Both groups received single‐surface phototherapy with four blue compact fluorescent tube lights. The light source was kept at a 20‐centimetre distance from the neonate. Neonates in both groups received exclusive breastfeeding. The study describes the feeding schedule of the turning group in the description of the intervention. However, the feeding schedule of the supine group is not described. Baseline characteristics like gestational age, birth weight, spectral irradiance (microwatt/cm²/nm), breastfeeding, Apgar score, weight loss, and STB at the start of phototherapy were comparable in the two groups. Outcomes reported included duration of phototherapy and absolute levels of STB at 12 hours of starting phototherapy.
Shinwell 2002 enrolled healthy term infants with birth weight above 2500 g and STB concentration above 18 mg/dL at the start of phototherapy. Of note, this study also included neonates with haemolytic jaundice. Neonates were randomised to supine (n = 16) or turned (n = 16) position. However, two neonates were later excluded from the turning group because of protocol violations: one received double‐surface phototherapy, and another underwent exchange transfusion. Both the study groups received single‐surface phototherapy with two blue and two white compact fluorescent tube lights. The light source was kept at a distance of 23 to 25 cm from the neonate. In the supine group, neonates were positioned supine only and received a 30‐minute break for feeding and nursing care every 150 minutes. In the turning group, neonates were positioned alternately supine or prone every 180 minutes (150 minutes of phototherapy and 30 minutes for feeding and nursing care). Baseline variables like birth weight, gestational age, gender, feeding type, age at initiation of phototherapy, haematocrit, and bilirubin at the start of phototherapy, and incidence of positive Coombs' test, were comparable in the two groups. Reported outcomes included the duration of phototherapy, absolute levels and fall of STB at 24 hours of starting phototherapy and at the end of phototherapy.
Further details of the included studies are provided in the Characteristics of included studies section.
Excluded studies
We excluded six identified articles from the review (Caldera 1984; Fakhraee 2011; Lee Wan Fei 2015; Pritchard 2004; Stanley 2004; Yamauchi 1989). Two studies had non‐random participant allocation (Fakhraee 2011; Yamauchi 1989); one study was a review (Lee Wan Fei 2015); and one study did not fit into inclusion criteria and covered other issues related to neonatal hyperbilirubinaemia (Stanley 2004). Another study tested a different intervention (partly clothed versus naked neonates) and was excluded (Pritchard 2004). The study by Caldera 1984 reported the results of two prospective surveys on phototherapy for neonatal jaundice and evaluated the influence of the intensity of the irradiance, mode and continuous or discontinuous phototherapy, and hence was excluded.
Further details are provided in the Characteristics of excluded studies section.
Studies awaiting classification
One study is awaiting classification (IRCT201111063250N4).
Further details are provided in the Characteristics of studies awaiting classification section.
Risk of bias in included studies
Ratings of the methodological quality of the included studies and risk of bias assessments are presented in the Characteristics of included studies section and are summarised in Figure 2 and Figure 3.

Risk of bias graph: review authors' judgements about each risk of bias item.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
Sequence generation
Two studies did not describe the method of sequence generation and were therefore at unclear risk of selection bias (Chen 2002; Mohammadzadeh 2004). Bhethanabhotla 2013 used a computer‐generated random sequence and therefore also had a low risk of selection bias. The other two studies described the use of random picking up of a sealed envelope to decide the study group allocation, which is likely to have been an acceptable method of sequence generation.
Allocation concealment
Three studies described the use of sealed envelopes. Bhethanabhotla 2013 used serially numbered, sealed envelopes, whilst Donneborg 2010 and Shinwell 2002 describe random picking up of a sealed envelope. The other two studies did not describe the use of any method to ensure allocation concealment and were therefore at unclear risk of selection bias.
Blinding
Blinding of participants and personnel
Given the nature of the intervention, blinding of healthcare providers or parents was not possible in any of the included studies.
Blinding of outcome assessors
None of the studies described any method to ensure blinding of outcome assessors. However, given the nature of the intervention and the role of the clinical care team in deciding important outcomes like duration of phototherapy, blinding of outcome assessors would not have been possible in any of the included studies.
Incomplete outcome data
Four studies described all the study outcomes for all the randomised neonates (Bhethanabhotla 2013; Chen 2002; Donneborg 2010; Mohammadzadeh 2004). However, in the study by Shinwell 2002, two enrolled neonates received a higher level of intervention (double‐surface phototherapy instead of single‐surface phototherapy) and were excluded from the analysis. The use of more effective co‐intervention could have been due to the failure of the treatment being investigated, therefore we assessed this study as at high risk of attrition bias.
Selective reporting
Bhethanabhotla 2013 reported all of the outcomes mentioned in the clinical trial registry and was therefore at low risk of reporting bias. The trial registry information was not available for the study by Chen 2002; however, all important outcomes were reported, therefore this study was also assessed as at low risk of reporting bias. In the study by Donneborg 2010, only information about the per cent fall of serum bilirubin and absolute values of serum bilirubin was provided, with no information provided regarding the duration of hospital stay and the need for an exchange transfusion. Due to the lack of a published protocol in a trial registry, it cannot be deduced whether all the predefined outcomes have been reported, therefore the study was assessed as having an unclear risk of reporting bias. For the two remaining studies, the trial registration information or study protocol was not available, and some important outcomes have not been reported, therefore these two studies were also assessed as having an unclear risk of reporting bias.
Other potential sources of bias
We could not assess the likelihood of potential publication bias using funnel plots, as at least 10 trials were not available.
Effects of interventions
See summary of findings Table 1 for the main comparison.
Periodic change in body position compared to only supine body position for neonates under phototherapy for the treatment of hyperbilirubinaemia
Duration of phototherapy
Four studies reported this outcome. Pooled results showed no significant difference was observed in the duration of phototherapy (mean difference (MD) 1.71 hours, 95% confidence interval (CI) −3.17 to 6.59 hours; 4 studies, 231 participants; Analysis 1.1). Statistical heterogeneity was significant (Chi² = 7.10, df = 3 (P = 0.07); I² = 58%). Overall, we judged that a periodic change in body position results in little to no difference in duration of phototherapy, based on low certainty evidence due to risk of bias in the included studies and significant heterogeneity.
Per cent fall in bilirubin at 24 hours of starting phototherapy
Three studies reported this outcome. Shinwell 2002 reported a significantly lower per cent fall of bilirubin at 24 hours of starting phototherapy (MD −8%, 95% CI −14.5% to −1.5%) in the supine study group. The remaining two studies did not report any significant difference between groups (Chen 2002; Donneborg 2010). Of the five included studies, only the study by Shinwell 2002 included neonates with haemolytic jaundice. However, the serum bilirubin levels at initiation of phototherapy were not higher than observed in the other included studies.
On pooling of the results, no significant difference was observed in the per cent fall in bilirubin at 24 hours of starting phototherapy (MD −1.17%, 95% CI −6.44% to 4.10%; 3 studies, 193 participants; Analysis 1.2). Statistical heterogeneity was significant (Chi² = 6.55, df = 2 (P = 0.04); I² = 69%). We downgraded the certainty of evidence for this outcome to low due to risk of bias in the included studies and significant heterogeneity.
Rate of fall of serum total bilirubin at 24 hours of starting phototherapy
Only one study reported this outcome (Bhethanabhotla 2013). The rate of fall of STB at 24 hours of starting phototherapy measured was not significantly different between groups (MD 0.02 mg/dL/h, 95% CI −0.02 to 0.06 mg/dL/h; 1 study, 100 participants; Analysis 1.3). This outcome is related to the per cent fall in serum bilirubin (Analysis 1.2). We downgraded the certainty of evidence for this outcome to low due to risk of bias in the included studies and imprecision.
Serum total bilirubin at 24 hours of starting phototherapy
Four studies reported this outcome. On pooling of the results, no significant difference was observed in STB at 24 hours of starting phototherapy (MD 0.22 mg/dL, 95% CI −0.43 to 0.86 mg/dL; 4 studies, 292 participants; Analysis 1.4). Statistical heterogeneity was not significant (Chi² = 3.95, df = 3 (P = 0.27); I² = 24%). Overall, we judged the certainty of evidence that a periodic change in body position results in little to no difference in STB 24 hours after starting phototherapy to be moderate due to risk of bias in the included studies.
Other outcomes
None of the studies reported important secondary outcomes like the need for or number of exchange transfusions, the incidence of bilirubin‐induced neurological damage (BIND), side effects of phototherapy, kernicterus, and sudden infant death syndrome (SIDS).
Subgroup analysis
Only one study included neonates with haemolytic jaundice (Shinwell 2002). However, separate data for neonates with haemolytic jaundice were not reported. In two studies, preterm neonates were eligible to be enrolled (Bhethanabhotla 2013; Donneborg 2010). However, separate data for preterm neonates were not reported. We therefore did not conduct the planned subgroup analyses comparing haemolytic versus non‐haemolytic jaundice and term versus preterm neonates.
Discussion
Summary of main results
Evidence from five studies in 343 term and preterm neonates indicates that a periodic change of body position under phototherapy as compared to no prescribed change may result in little or no difference in the duration of phototherapy and rate of fall in bilirubin at 24 hours of starting phototherapy. The available evidence is insufficient to conclusively answer if a periodic change in comparison to no prescribed change of body position improves the efficacy of phototherapy. The certainty of the evidence for the review outcomes was low due to risk of bias in the included studies, imprecision, or heterogeneity. For important review outcomes such as duration of phototherapy, per cent fall in bilirubin at 24 hours, and rate of fall of bilirubin, the effect estimates were too imprecise to determine their clinical importance. The 95% CIs of absolute measures of effectiveness cross the boundaries of clinically important effect size. The duration of phototherapy could be 3.17 hours shorter to 6.59 hours longer when a periodic change in body position is compared with no prescribed change in position. Similarly, the rate of fall of bilirubin at 24 hours of starting phototherapy could be 0.02 mg/dL/h lower to 0.06 mg/dL/h higher. None of the included studies reported on the need for or number of exchange transfusions, incidence of BIND, and SIDS.
Overall completeness and applicability of evidence
Periodic change in body position probably has little or no effect on the rate of fall of serum total bilirubin at 24 hours of starting phototherapy. Results of this systematic review apply mainly to neonates born at late‐preterm or term gestation receiving phototherapy for non‐haemolytic hyperbilirubinaemia. Only one included study enrolled neonates with haemolytic jaundice (Shinwell 2002). The results of these infants were not reported separately; however, the study results by Shinwell 2002 suggest that turning of term infants with hyperbilirubinaemia does not increase the efficacy of phototherapy, rather that infants nursed supine had a significantly greater drop in bilirubin and shorter duration of phototherapy. We did not find any specific elements in study design or conduct that could account for this, although this was the only included study that enrolled neonates with haemolytic hyperbilirubinaemia. It is possible that repeated interruptions in the phototherapy to change position of the baby could have interfered with efficacy of the phototherapy. Preterm neonates (≥ 35 weeks' and ≥ 33 weeks' gestation, respectively) were eligible to be enrolled in two of the five studies included in the review (Bhethanabhotla 2013; Donneborg 2010). However, separate data for preterm neonates were not reported. Of note, the effect of frequent change in body position is relevant only if the neonate is receiving single‐surface phototherapy. The potential effect of change in body position may be reduced if double‐surface phototherapy is being delivered with two light sources ‐ one over the head and the second underneath with neonate lying on a fibreoptic phototherapy blanket or on a glass surface covering the phototherapy lights underneath the neonate. Lastly, the rationale for changing position during phototherapy rests on the assumption that the effects of phototherapy are largely limited to bilirubin residing in the superficial and connective tissues of the skin, whereas it may be that the effects of phototherapy on bilirubin in the capillary circulation are also important (Hansen 2010).
Any recommendation regarding change in body position of neonates needs to take into account the association of prone positioning with increased risk of SIDS. Supine position is recommended for neonates when put to sleep at home. However, in hospitals, preterm neonates, especially those with respiratory distress, are frequently nursed in a prone position. Whilst during intensive care and continuous monitoring prone positioning is practised for benefits in oxygenation and as part of developmentally supportive care, its safety when providing phototherapy in less intensive areas of the hospital including postnatal wards and rooms or during home phototherapy has not been evaluated. None of the five studies included in this review reported the outcome of SIDS. It is unlikely that a study would ever be conducted to evaluate this outcome during phototherapy, as the potential benefits to improve the effectiveness of phototherapy are likely to be small, and such a trial would need to enrol thousands of neonates.
Quality of the evidence
We judged the included studies to be at high risk of bias. The overall certainty of the evidence was low to very low due to statistical heterogeneity and unclear risk of selection bias (summary of findings Table 1). The main reasons for downgrading the evidence included unclear reporting, statistical heterogeneity, imprecision, and unclear risk of selection bias (two studies). Blinding of outcome assessors was not possible in any of the included studies.
Potential biases in the review process
We did not search Embase or ClinicalTrials.gov separately. Therefore, the retrieval of available studies on phototherapy may have been reduced. While trial records from Embase and ClinicalTrials.gov are included in CENTRAL, we acknowledge a publishing delay from when study records are first available in their original sources and when they are available in CENTRAL. This publishing delay may have prevented recent trial records and reports from being identified. Further, searching only CENTRAL for records from these sources may have lessened the likelihood of retrieving eligible studies due to the limited number of fields that are published to CENTRAL compared to the original source databases. For future updates of this review, we will search Embase and ClinicalTrials.gov separately to ensure maximum retrieval of eligible study records.
It is unlikely that the literature search strategy used in this review would have missed any relevant randomised controlled trials. One trial is awaiting classification (IRCT201111063250N4); although completed, the results have not been published in the literature or presented at any conference. We did not exclude any other potentially relevant study, and there were no marginal decisions affecting the inclusion of any study in the review. However, due to the limited number of eligible studies, we were not able to evaluate publication bias. We have used the standard methods recommended by Cochrane Neonatal to minimise the risk of bias in conducting this review. Two review authors independently assessed the eligibility of studies for inclusion in the review, extracted data, and assessed risk of bias. We could not perform subgroup analysis due to a lack of separate and adequate data in haemolytic hyperbilirubinaemia and in preterm neonates.
Agreements and disagreements with other studies or reviews
Lee Wan Fei and Abdullah performed a systematic review on the effect of turning versus supine position under phototherapy on neonates with hyperbilirubinaemia (Lee Wan Fei 2015). They included five studies identical to the current review, although meta‐analysis was not performed. The authors presented the findings of individual studies and concluded that both supine and turning position were equally effective during phototherapy. Lee Wan Fei 2015 excluded studies not available in the English language and studies with position changes more frequent than two‐ to three‐hourly. We did not apply any language restriction in our search strategy. However, we did not find any non‐English study in our search. We did not apply any time restriction in the change of position in selecting studies for inclusion in the current review.

Study flow diagram.

Risk of bias graph: review authors' judgements about each risk of bias item.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.

Comparison 1: Turning position versus supine under phototherapy, Outcome 1: Duration of phototherapy

Comparison 1: Turning position versus supine under phototherapy, Outcome 2: Per cent fall in bilirubin at 24 hours of starting phototherapy

Comparison 1: Turning position versus supine under phototherapy, Outcome 3: Rate of fall of serum total bilirubin at 24 hours of starting phototherapy

Comparison 1: Turning position versus supine under phototherapy, Outcome 4: Serum total bilirubin at 24 hours of starting phototherapy
| Turning position versus supine under phototherapy in term and preterm neonates with hyperbilirubinaemia | ||||||
| Patient or population: term and preterm neonates with hyperbilirubinaemia | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | No. of participants | Certainty of the evidence | Comments | |
|---|---|---|---|---|---|---|
| Risk with turning position under phototherapy | Risk with supine position under phototherapy | |||||
| Duration of phototherapy (hours) | The mean duration of phototherapy ranged from 24.8 to 52.8 hours. | The mean duration of phototherapy ranged from 25.5 to 53.3 hours. | Mean duration of phototherapy was on average 1.71 hours longer in the turning group (95% CI −3.17 to 6.59). | 231 | ⊕⊕⊝⊝ | Periodic change in body position may lead to little or no difference in the duration of phototherapy. |
| Rate of fall of serum total bilirubin at 24 hours of starting phototherapy (mg/dL/h)b | The mean rate of fall of serum total bilirubin at 24 hours of starting phototherapy was 0.22 mg/dL/h. | The mean rate of fall of serum total bilirubin at 24 hours of starting phototherapy was 0.2 mg/dL/h. | MD in the rate of fall of serum total bilirubin was an average of 0.02 mg/dL/h higher in the turning group | 100 | ⊕⊕⊝⊝ Lowc | Periodic change in body position may lead to little or no effect on the rate of fall of serum total bilirubin at 24 hours of starting phototherapy. |
| Need for exchange transfusion | None of the studies reported this outcome. | |||||
| Bilirubin‐induced neurological damage (BIND) | None of the studies reported this outcome. | |||||
| Sudden infant death syndrome (SIDS) | None of the studies reported this outcome. | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| aDowngraded by two levels because of statistical heterogeneity and unclear risk of selection bias. (One study has attrition bias. Three studies have unclear reporting. Blinding of outcome assessors was not possible in any of the studies.) | ||||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1.1 Duration of phototherapy Show forest plot | 4 | 231 | Mean Difference (IV, Random, 95% CI) | 1.71 [‐3.17, 6.59] |
| 1.2 Per cent fall in bilirubin at 24 hours of starting phototherapy Show forest plot | 3 | 193 | Mean Difference (IV, Random, 95% CI) | ‐1.17 [‐6.44, 4.10] |
| 1.3 Rate of fall of serum total bilirubin at 24 hours of starting phototherapy Show forest plot | 1 | 100 | Mean Difference (IV, Random, 95% CI) | 0.02 [‐0.02, 0.06] |
| 1.4 Serum total bilirubin at 24 hours of starting phototherapy Show forest plot | 4 | 292 | Mean Difference (IV, Random, 95% CI) | 0.22 [‐0.43, 0.86] |

