Oxigenoterapia en ámbitos prehospitalarios para pacientes con una exacerbación aguda de la enfermedad pulmonar obstructiva crónica
Resumen
Antecedentes
La enfermedad pulmonar obstructiva crónica (EPOC) es una causa principal de morbilidad y mortalidad a nivel mundial, caracterizada por el deterioro agudo de los síntomas. Durante estas exacerbaciones, los pacientes son propensos a desarrollar hipoventilación alveolar, que puede ser causada en parte por la administración de concentraciones altas de oxígeno inspirado.
Objetivos
Determinar el efecto de diferentes concentraciones de oxígeno inspirado ("flujo alto" en comparación con "controlado") en el ámbito prehospitalario (antes del departamento de urgencias) sobre los resultados de los pacientes con exacerbaciones agudas de la EPOC (EAEPOC).
Métodos de búsqueda
Se realizaron búsquedas en el Registro Especializado del Grupo Cochrane de Vías Respiratorias (Cochrane Airways Group Specialised Register), en las listas de referencias de los artículos y en las bases de datos de ensayos clínicos en línea. También se estableció contacto con los autores de los ensayos controlados aleatorizados (ECA) identificados para obtener detalles de otros estudios relevantes publicados y no publicados. La búsqueda más reciente se realizó el 16 de septiembre 2019.
Criterios de selección
Se incluyeron ECA que comparaban la oxigenoterapia a diferentes concentraciones o la oxigenoterapia versus placebo en el ámbito prehospitalario para el tratamiento de las EAEPOC.
Obtención y análisis de los datos
Dos autores de la revisión evaluaron de forma independiente la calidad de los ensayos y extrajeron los datos. El resultado primario fue la mortalidad por todas las causas y relacionada con la respiración.
Resultados principales
La búsqueda identificó un total de 824 citas; se identificó un estudio para su inclusión y dos estudios están a la espera de clasificación. Los 214 participantes del estudio incluido eran adultos con EAEPOC, que recibieron tratamiento por parte de los paramédicos en camino al hospital. La media de edad de los participantes era de 68 años.
Se observó una reducción de la mortalidad prehospitalaria e intrahospitalaria a favor del grupo de oxígeno en dosis ajustada (dos muertes en el grupo de oxígeno en dosis ajustada en comparación con 11 muertes en el brazo de control de flujo alto; riesgo relativo (RR) 0,22; intervalo de confianza (IC) del 95%: 0,05 a 0,97; 214 participantes). Estas cifras se traducen en un efecto absoluto de 94 por 1000 (oxígeno de flujo alto) comparado con 21 por 1000 (oxígeno en dosis ajustada), y un número necesario a tratar para lograr un resultado beneficioso adicional (NNTB) de 14 (IC del 95%: 12 a 355) con la oxigenoterapia en dosis ajustada. Aparte de la mortalidad, no se informó de ningún otro evento adverso en el estudio incluido.
Se observaron intervalos de confianza amplios entre los grupos para los gases en sangre arterial (lo cual puede presentar factores de confusión debido a la infidelidad en cuanto al protocolo en el estudio incluido para esta medida de resultado), el fracaso del tratamiento que requiere ventilación invasiva o no invasiva o la utilización del hospital. No se informaron datos para la calidad de vida, la función pulmonar o la disnea. El riesgo de sesgo dentro del estudio incluido fue en gran parte incierto, aunque hubo un riesgo alto de sesgo en los dominios relacionados con el sesgo de realización y de deserción. Según los criterios GRADE la evidencia se consideró de certeza baja.
Conclusiones de los autores
El único estudio incluido encontró una reducción de la mortalidad prehospitalaria e intrahospitalaria en el brazo de oxígeno en dosis ajustada en comparación con el brazo de control de flujo alto. Sin embargo, la escasez de evidencia limita un poco la fiabilidad de estos hallazgos y la posibilidad de generalizarlos a otros ámbitos. Se necesitan ECA sólidos y bien diseñados para investigar más a fondo el efecto de las oxigenoterapias en el ámbito prehospitalario para los pacientes con EAEPOC.
PICO
Resumen en términos sencillos
Oxigenoterapia en el ámbito prehospitalario para el empeoramiento súbito de la enfermedad pulmonar obstructiva crónica
Antecedentes
La enfermedad pulmonar obstructiva crónica (EPOC) es una enfermedad crónica caracterizada por el empeoramiento progresivo de la reducción del flujo de aire a través de los pulmones. Los pacientes con EPOC son propensos a episodios súbitos en los que sus síntomas empeoran (p.ej. aumento persistente de la dificultad respiratoria y cambios en el volumen y consistencia de las mucosas) y los niveles de oxígeno pueden disminuir. El tratamiento inicial durante estos episodios, cuando los pacientes son transportados al hospital, por lo general incluye oxígeno. Sin embargo, la administración de demasiado oxígeno a estos pacientes puede causar un aumento en el nivel de dióxido de carbono que eventualmente puede dar lugar a una reducción de la frecuencia respiratoria y posiblemente detener de forma completa su capacidad de respirar.
Pregunta de la revisión
Esta revisión se propuso investigar si la oxigenoterapia administrada en diferentes concentraciones sobre la base de las necesidades de los pacientes sería menos perjudicial, más perjudicial o no marcaría ninguna diferencia en comparación con un grupo de control que utiliza oxígeno de flujo alto.
Características de los estudios
Para investigar esta pregunta se realizaron búsquedas de ensayos controlados aleatorizados (ECA), que son estudios en los que las personas involucradas tienen probabilidades iguales de recibir el tratamiento o el comparador. El interés se centró en los ensayos que comparaban diferentes tasas de flujo (concentraciones) de oxígeno administrado en una ambulancia a los pacientes trasladados al hospital debido al empeoramiento súbito de los síntomas de la EPOC.
Resultados clave
Solo se encontró un estudio que consideró la pregunta de la revisión. El estudio asignó al azar a los participantes para recibir oxígeno en dosis ajustada (oxigenoterapia administrada en diferentes concentraciones adaptadas a las necesidades del paciente con el fin de mantener los niveles de oxígeno en la sangre entre el 88% y el 92%) u oxígeno de flujo alto (oxigenoterapia administrada en una concentración consistentemente alta).
Hubo menos muertes (dos pacientes) en el grupo que recibió oxígeno en dosis ajustada, en comparación con el grupo de control que utilizó oxígeno de flujo alto administrado a ocho a diez litros por minuto con una mascarilla (11 pacientes).
Certeza de la evidencia
Debido a la inclusión de un único estudio y al número reducido de muertes que ocurrieron, la confianza en el tamaño de la diferencia entre los dos tratamientos es limitada. Se consideró que la evidencia era de certeza baja.
Conclusión
El único estudio incluido encontró que la administración de concentraciones de oxígeno adaptadas individualmente a los pacientes cuando son transportados al hospital con un empeoramiento súbito de la EPOC, reduce el riesgo de muerte en comparación con el uso de una concentración de oxígeno consistentemente alta. Sin embargo, el conjunto de evidencia es demasiado pequeño para afirmar con seguridad que el oxígeno en dosis ajustada es menos perjudicial y más efectivo que el oxígeno de flujo alto en este grupo de pacientes en general.
Este resumen en términos sencillos está actualizado hasta septiembre 2019.
Authors' conclusions
Summary of findings
| Titrated oxygen therapy compared to high‐flow oxygen therapy for acute exacerbations of chronic obstructive pulmonary disease | ||||||
| Patient or population: adults with acute exacerbations of chronic obstructive pulmonary disease | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | № of participants | Certainty of the evidence | Comments | |
| Risk with high‐flow oxygen therapy | Risk with titrated oxygen therapy | |||||
| Mortality (respiratory‐ related and all‐cause | 94 per 1,000 | 21 per 1,000 | RR 0.22 | 214 | ⊕⊕⊝⊝ | A difference in mortality was observed, with 11 deaths in the high‐flow oxygen arm compared to two deaths in the titrated oxygen arm (P = 0.05). This translates to a number needed to treat for an additional beneficial outcome (NNTB) of 14 (95% CI 12 to 355) with administration of titrated oxygen therapy, and is shown as a Cates plot in Figure 1. All deaths occurred after arrival at the hospital; two were in intensive care. Respiratory failure was the cause of mortality in all cases, with approximately 70% of deaths occurring within the first five days following admission for both treatment arms. |
| Arterial blood gas (pH) | The mean arterial blood gas (pH) was 7.29 | MD 0.06 pH higher | ‐ | 214 | ⊕⊕⊝⊝ | Based on the intention‐to‐treat analysis for the COPD subgroup, no significant difference between treatment arms for blood gas measurements was observed between groups (P = 0.23). Only 11% of participants had this measurement performed according to protocol. |
| Ventilation of any type | 143 per 1,000 | 96 per 1000 | RR 0.67 | 189 | ⊕⊕⊝⊝ | No significant difference observed between treatment arms for ventilation requirement for per protocol or intention‐to‐treat analyses. |
| Length of hospital stay | The mean length of hospital stay was 6.3 days | MD 0.88 days lower | ‐ | 214 | ⊕⊕⊝⊝ | No significant difference was observed between treatment arms in length of hospital stay for the intention‐to‐treat analysis (P = 0.21). |
| Quality of life ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| Lung function ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| Dyspnoea score ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded once for indirectness because there was a single included study with moderate sample size (n = 214). The study was conducted in one state within Australia only. | ||||||

In the high‐dose oxygen group 9 people out of 100 died, compared to 2 (95% confidence interval 0 to 9) out of 100 for the titrated oxygen group.
Background
This is an update of a Cochrane Review previously published in 2006 (Austin 2006).
Description of the condition
Chronic obstructive pulmonary disease (COPD) is the fourth leading cause of death worldwide (Lozano 2013) and contributes to global morbidity and mortality, particularly in those over 45 years of age. This burden is expected to continue to rise for the foreseeable future. By 2020, COPD is expected to be the third leading cause of death in the world (GOLD 2018; Lozano 2013; Mendis 2014; Vos 2012). COPD is not one single condition, but instead is an umbrella term used to describe a combination of chronic lung diseases that cause inflammation, including chronic bronchitis and emphysema (WHO 2018). Tobacco smoking is known to be the most common cause of COPD; lifelong smokers having a 50% probability of developing COPD (Laniado‐Laborin 2009). Other modifiable risk factors include: exposure to environmental smoke (such as fumes from carbon‐based cooking, including early childhood exposure, and heating fuels used for cooking like charcoal and gas), occupational hazards (such as exposure to chemicals and pollutants), poor nutrition, intrauterine exposures and pneumonia or other respiratory infections during childhood (AIHW 2016). Non‐modifiable risk factors include age (COPD is more common as people age), low socioeconomic status index and genetic predisposition (primarily alpha‐1 antitrypsin deficiency) (AIHW 2016; Grigsby 2016).
Chronic obstructive pulmonary disease is characterised by irreversible and progressive destruction of lung parenchyma and structural change to small airways owing to a chronic inflammatory process (GOLD 2018). This results in breathlessness (initially with exertion), chronic cough and sputum production that gradually worsen as the condition progresses over time (WHO 2018). An acute, abnormal deterioration of respiratory symptoms is termed an acute exacerbation of COPD (AECOPD) (GOLD 2018). These are serious episodes of increasing breathlessness, cough and sputum production that can last from several days to weeks; they can require hospitalisation and result in death (WHO 2018). COPD patients may develop alveolar hypoventilation, which results in an elevation of the partial pressure of arterial carbon dioxide (PaCO₂), known as hypercarbia, and a reduction in partial pressure of arterial oxygen (PaO₂), known as hypoxaemia (Abdo 2012). The combination of a PaO₂ below 60 and PaCO₂ above 45 is referred to as hypoventilatory (hypercapnic) respiratory failure. It is associated with poor outcomes in AECOPD (Groenewegen 2003). As COPD is a heterogenous condition, different pathological processes predominate in different areas of the lung, resulting in wasted perfusion and differentially oxygenated blood on return to systemic circulation (Brill 2014; Cooper 2008).
Description of the intervention
In healthy non‐smoking adults, oxygen saturations of 94% to 98% are considered normal, with levels diminishing as people age (Brill 2014; Kane 2013). For example, a 70 year old may have an oxygen saturation of less than 94%, which may still be considered normal, particularly in the presence of comorbidities such as heart failure (Kane 2013). People having an AECOPD typically present to hospital with lower levels of oxygen saturation in the blood due to the destruction of lung parenchyma and structural changes resulting in poor gas exchange (GOLD 2018). Thus, administration of oxygen at concentrations greater than that found in the surrounding air will help to increase oxygen saturation or carbon dioxide excretion, or both (Brill 2014). Oxygen is one of the most commonly administered drugs in the pre‐hospital and emergency setting; 34% of people being transported by ambulance are estimated to receive it (Hale 2008). Thus administration in the pre‐hospital setting typically occurs via medical or paramedical assistance, such as during ambulance transport. According to current guidelines its administration should be delivered with a targeted oxygen saturation rate and regular monitoring of patient response, though this is based largely on single studies and expert opinion (Beasley 2015). In the context of COPD, controlled supplemental oxygen (rather than high‐flow oxygen) is typically adequate to overcome hypoxia related to an AECOPD and avoid respiratory acidosis and hypercapnic respiratory failure (Brill 2014; O'Driscoll 2017). The amount of oxygen delivered to an individual can be described in a number of ways. Most commonly it is described as a flow rate, which refers to the number on the dial the health professional selects on the oxygen flow metre; or fraction of inspired oxygen (FiO2), which denotes the concentration of oxygen inhaled by the individual and is expressed as a percentage. A recent subgroup analysis has indicated that administration of six litres of oxygen per minute to people with COPD, who had a normal concentration of oxygen in their blood and suspected acute myocardial infarction, was of no benefit (Andell 2019). Non‐experimental studies indicate increased mortality with high‐flow oxygen therapy in the hospital and pre‐hospital setting (Denniston 2002; Wijesinghe 2011). Hence, a target SpO₂ (arterial oxygen saturation measured by pulse oximeter) range of 88% to 92% for the treatment of AECOPD is recommended in guidelines from Australia (Beasley 2015), Europe (Kane 2013), the UK (O'Driscoll 2017) and America (Rochwerg 2017). Oxygen can be administered via nasal cannulae or face masks, with nasal cannulae reported to be the simplest mode of administration as they are less likely to fall off and allow patients to speak while undergoing treatment (Brill 2014). Disadvantages to nasal cannulae, however, include patient reports of nasal dryness/epistaxis and discomfort as well as ineffectiveness due to mouth breathing, degree of nasal congestion and respiratory rate and/or minute volume (Brill 2014).
How the intervention might work
Administration of oxygen to people with COPD is intended to relieve hypoxaemia and as a mainstay of treatment for exacerbations is often delivered in conjunction with other treatments, including inhaled bronchodilators (Beasley 2015; Kane 2013; O'Driscoll 2017). Impairment of gas exchange and the development of respiratory failure may be precipitated by a variety of factors, one of which is the administration of oxygen at high concentrations. In this group of patients, the administration of high inspired concentrations of oxygen may cause a worsening of respiratory function, leading to further elevation of arterial carbon dioxide concentration (Donald 1949; McNicol 1965; Plant 2001). In addition, high‐flow oxygen may cause injury to other organs, such as the heart, and lead to morbidity and mortality through other mechanisms. Therefore, it is important for oxygen therapy to be controlled with monitoring to overcome hypoxia and prevent oxygen‐induced hypercapnia, according to patient needs (Brill 2014). Oxygen titration can be achieved through altering the oxygen flow rate or administering a mixture of air and oxygen in set proportions, with certain delivery devices, to allow the patient to breathe a known fraction of inspired oxygen (Brill 2014). When the appropriate level of oxygen is administered to reach the target SpO₂ of 88% to 92% in the pre‐hospital setting, this will relieve hypoxaemia and reduce the risk of mortality (Brill 2014).
Why it is important to do this review
Supplemental oxygen therapy in the emergency situation has traditionally been administered using "high‐flow" oxygen. This approach has been adopted on the basis that the population as a whole are at greater risk from failure to correct oxygenation than from its excessive administration. Indeed, for people with AECOPD, nebulised bronchodilators are often administered through a mask using oxygen at flows of 6 L/min to 8 L/min. A retrospective study found that the administration of an inspired oxygen concentration above 28% was associated with a 14% in‐hospital mortality rate, compared to 2% in‐hospital mortality for those administered a concentration of oxygen less than 28% (Denniston 2002). This has raised questions about the optimal delivery of oxygen to people with AECOPD in the pre‐hospital setting. One prospective prevalence study in the UK identified that one in five patients out of 983 admitted to hospital with AECOPD had respiratory acidosis (Plant 2000). A more recent cohort study of 415 patients with AECOPD conducted across Asia, Australia and New Zealand identified low compliance with blood gas testing, particularly in South East Asia, suggesting the potential for under‐diagnosis of clinically important hypercapnia, which may have implications for oxygen therapy (Kelly 2018). Therefore, the potential for high‐flow oxygen administration in the pre‐hospital setting, coupled with lack of blood gas testing to identify acidosis in the emergency department, presents a need for evidence consolidation around the application of oxygen therapy in the pre‐hospital setting. The results of this review will provide updated evidence for clinical practice guidelines and raise awareness of this important clinical issue.
Objectives
To determine the effect of different inspired oxygen concentrations ("high flow" compared to "controlled") in the pre‐hospital setting (prior to casualty/emergency department) on outcomes for people with acute exacerbations of COPD.
Methods
Criteria for considering studies for this review
Types of studies
We considered randomised controlled trials (RCTs) for inclusion within this review, with full text and abstracts both accepted. There were no restrictions on language or publication date.
Types of participants
Studies were included that evaluated adults with AECOPD requiring medical or paramedical assistance in the pre‐hospital setting. AECOPD was defined as any combination of: an increase in breathlessness, sputum volume, sputum purulence, cough or wheeze. COPD itself was defined as spirometric evidence of airflow obstruction (forced expired volume in one second (FEV₁)/forced vital capacity (FVC) less than 0.7) plus a smoking history of greater than 10 pack history years; or disease matching an accepted standard definition (e.g. Global initiative for chronic Obstructive Lung Disease (GOLD) or American Thoracic Society (ATS)). Studies were excluded if they investigated patients with acute asthma.
Types of interventions
Studies were included that evaluated high fractional inspired concentration (FiO₂) (greater than 28%) oxygen therapy compared to either: 1) placebo; 2) low FiO₂ (less than 28%) oxygen therapy; 3) titrated oxygen therapy; or 4) standard care.
In acknowledgement that oxygen therapy is rarely seen in isolation when treating AECOPD, additional medical treatment interventions (e.g. bronchodilators and corticosteroids) were accepted but were required to be standardised to both groups as far as possible.
Types of outcome measures
Primary outcomes
Mortality (respiratory‐related and all‐cause)
Secondary outcomes
-
Quality of life
-
Arterial blood gas
-
Lung function (FEV₁/FVC)
-
Dyspnoea score (as measured by accepted clinical scales: Medical Research Council scale, Borg, Chronic Respiratory Disease Questionnaire, Baseline Dyspnoea Index, St. George's Respiratory Questionnaire)
-
Treatment failure requiring intensive care unit (ICU), invasive or non‐invasive ventilation
-
Hospital utilisation (length of stay, readmission rate, emergency department presentations)
Reporting in the publications of one or more of the outcomes listed above was not an inclusion criterion for the review.
Search methods for identification of studies
Electronic searches
The previously published version of this review included searches up to August 2008 and did not find any studies for inclusion. The search period for this update is August 2008 to September 2019. For this update we searched the Cochrane Airways Specialised Register of trials up to 16 September 2019 through contact with the Information Specialist for the Group, using search terms relevant to this review. The Cochrane Airways Specialised Register contains studies identified from several sources:
-
Monthly searches of the Cochrane Central Register of Controlled Trials (CENTRAL) via the Cochrane Register of Studies (CRS Web);
-
Weekly searches of MEDLINE Ovid SP;
-
Weekly searches of Embase Ovid SP;
-
Monthly searches of PsycINFO Ovid SP;
-
Monthly searches of CINAHL EBSCO (Cumulative Index to Nursing and Allied Health Literature);
-
Monthly searches of AMED EBSCO (Allied and Complementary Medicine);
-
Handsearches of the proceedings of major respiratory conferences.
Studies contained in the Specialised Register are identified through search strategies based on the scope of Cochrane Airways. Please see Appendix 1 for details of these strategies, as well as a list of handsearched conference proceedings. See Appendix 2 for search terms used to identify studies for this review.
A search of ClinicalTrials.gov (www.ClinicalTrials.gov) and the World Health Organization (WHO) trials portal (www.who.int/ictrp/en/) were also conducted. All sources were searched from inception, with no restriction on language of publication.
Searching other resources
We handsearched reference lists of all available included studies and review articles to identify potentially relevant citations and we made enquiries to authors of included studies regarding other published or unpublished trials known to them.
Data collection and analysis
Selection of studies
Two review authors (ZK and KVC) independently screened the titles and abstracts returned from the literature search and separated them into the following groups: 'potential inclusion' or 'exclude not relevant'. Full text was obtained where possible for the studies in the 'potential inclusion group' and the same two review authors independently assessed them for inclusion. Excluded studies were either tagged as 'exclude not relevant' or 'exclude but relevant', with reasons for exclusion recorded. The authors encountered no disagreement during the study selection process, however, had there been disagreements these would have been resolved in consultation with a third review author. A PRISMA flow diagram was used to demonstrate the study selection process.
Data extraction and management
Two review authors (ZK and KVC) independently extracted data for the included trial into a standardised, pilot tested data extraction form. Data extracted in duplicate included: study characteristics, outcome data and information on risk of bias. Any disagreements were resolved through discussion. One review author (KVC) entered the data into Review Manager 5 (Review Manager 2014) and these were double‐checked by ZK to ensure they were transferred correctly.
Assessment of risk of bias in included studies
In line with recommendations provided in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2011), two review authors (ZK and KVC) assessed the included study for risk of bias relating to random sequence generation, allocation concealment, blinding of participants and outcome assessors, handling of missing data, selective outcome reporting and other threats to study validity. An assessment of either high, unclear or low risk of bias was assigned to each domain with supporting justification recorded in the 'Risk of Bias' table. Disagreements during the 'Risk of bias' assessment process were resolved through discussion.
Assessment of bias in conducting the systematic review
This review has been revised to reflect current Cochrane reporting standards. All steps taken are described in the methods section of this publication. The original authors of this review (MA and RWB) are also the authors of the included study (Austin 2010); neither of them were involved in the extraction or interpretation of data from this study, this was solely completed by ZK and KVC.
Measures of treatment effect
Meta‐analysis was to be conducted where it would provide a meaningful outcome, e.g. where studies had sufficiently homogeneous methodology. Had there been enough studies for meta‐analysis, we would have extracted continuous and dichotomous data and analysed them in accordance with standard statistical techniques, with a random‐effects model for all studies deemed clinically and methodologically similar enough to be pooled. Had meta‐analysis been possible, mean differences (MDs) with 95% confidence interval (CIs), and pooled MDs for standardised mean differences (SMDs), would have been calculated for continuous outcomes. Risk ratios (RRs) with 95% CIs would have been calculated for dichotomous outcomes.
Given there was only a single study for inclusion, a qualitative synthesis was conducted and all data were entered and synthesised using Review Manager 5 software, according to the pre‐specified protocol. Despite no meta‐analysis being conducted the data for all reported, relevant outcomes were used to create individual forest plots for visualisation of the data.
Unit of analysis issues
The unit of analysis was considered to be the participant. Had there been sufficient studies for meta‐analysis, unit of analysis issues would have been addressed through the use of generic inverse variance and entering effect estimates and their standard errors, according to the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2019).
Dealing with missing data
Should there have been a need, missing information regarding participants on an available case analysis basis would have been conducted, as described in the Cochrane Handbook for Systematic Reviews of Interventions (Deeks 2019). If statistics essential for analysis were missing (e.g. when group means and standard deviations for both groups were not reported) and could not be calculated from other data, attempts would have been made to contact study authors to obtain missing data. In the event of loss of participants that occurred before baseline measurements were obtained, an assumption would have been made that this would have no effect on the final outcome.
Assessment of heterogeneity
We planned to assess statistical heterogeneity using a combination of methods, including visual inspection of data and use of the I² statistic. We would have judged an I² value of 50% or more to indicate the presence of substantial heterogeneity. Statistical significance would have been determined by the Der‐Simonian and Laird method of analysis presented with a P value less than 0.05 (Der Simonian 1986). We planned to investigate heterogeneity using pre‐specified subgroup analysis. Given no meta‐analyses were conducted in this review, heterogeneity was not assessed.
Assessment of reporting biases
We planned to explore potential reporting biases using a funnel plot had there been ten or more studies available for meta‐analysis. Instead, this was extrapolated as a possible risk of bias within the "other bias" section in the 'Risk of bias' tables.
Data synthesis
Data were combined from included trials using Review Manager 5 (Review Manager 2014) software. Studies were reported by using intention‐to‐treat analysis when all participants who were randomly assigned during the study were assessed, regardless of whether they received the intervention/study treatment to which they were allocated.
Summary of findings table
A 'Summary of findings table' was constructed from the available data, using the following outcomes.
-
Mortality
-
Quality of life
-
Arterial blood gas
-
Lung function (FEV₁/FVC)
-
Dyspnoea score
-
Treatment failure requiring intensive care unit (ICU), invasive or non‐invasive ventilation
-
Hospital utilisation (length of stay, readmission rate, emergency department presentations)
Quality of the body of evidence was assessed according to the following five GRADE considerations.
-
Study limitations
-
Indirectness
-
Inconsistency of results
-
Imprecision of results
-
Publication bias
This assessment was undertaken in accordance with guidelines from the Cochrane Handbook for Systematic Reviews of Interventions (Schünemann 2019), using GRADEpro GDT software (GRADEpro GDT). Justification for decisions regarding the upgrading or downgrading of evidence quality for each outcome was provided through footnotes.
Subgroup analysis and investigation of heterogeneity
Had there been sufficient studies for meta‐analysis, we planned to conduct the following subgroup analyses.
-
High fractional inspired concentration oxygen therapy versus placebo
-
High fractional inspired concentration oxygen therapy versus low fractional inspired concentration oxygen
-
High fractional inspired concentration oxygen therapy versus titrated oxygen therapy
-
High fractional inspired concentration oxygen therapy versus standard care
Sensitivity analysis
Had there been sufficient studies for meta‐analysis, we planned to conduct the following sensitivity analyses.
-
Excluding studies at high risk of bias in one or more domain
-
Comparing a random‐effects with a fixed‐effect model
Summary of findings and assessment of the certainty of the evidence
Results
Description of studies
For additional information, see: Characteristics of included studies, Characteristics of excluded studies and Characteristics of ongoing studies.
Results of the search
The previous version of this review did not identify any included studies. Through database searches conducted for this updated version, which covered the period from 2008 to 16 Septmber 2019, we identified a total of 854 records for screening. Thirty‐eight additional records were identified through screening of grey literature such as online clinical trial registries, producing a total of 889 records for review. On the basis of title and abstract, we excluded 794 records as they were not relevant. Full text was obtained for the remaining 30 records. Five records, representing one complete study and two ongoing studies, were found to be relevant and were included in the review. The remaining 23 records were assessed as excluded but relevant. For further details of screening processes, see the study flow diagram (Figure 2).
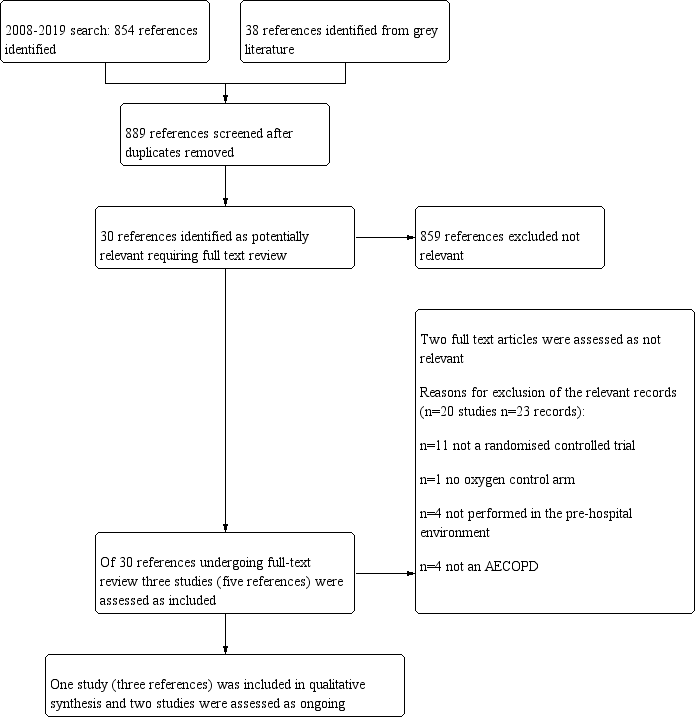
Study flow diagram.
Included studies
One study (three records) was identified for inclusion (Austin 2010). While this study enrolled 405 participants, only 214 of these had a diagnosis of AECOPD and it is this subset that has been included in analyses for this review. This study was a cluster‐randomised controlled, parallel‐group trial undertaken with the ambulance service in Hobart, Tasmania, Australia. The study population was comprised of people aged 35 years or older with breathlessness and a history or risk of COPD. They were treated by paramedics, transported and admitted to the Royal Hobart Hospital.
Intervention
Participants received titrated oxygen treatment delivered by nasal prongs to achieve arterial oxygen saturations between 88% and 92%, with concurrent bronchodilator treatment delivered by a nebuliser driven by compressed air and delivered via a facemask over the nasal prongs. Pulse oximeters were used to measure oxygen saturations and titrate oxygen to target saturations. All participants received other standard treatment according to Tasmanian Ambulance Service guidelines, including basic support, nebulised bronchodilators (salbutamol 5 mg made up in 2.5 mL normal saline, ipratropium bromide 500 μg made up with 2.5 mL normal saline), dexamethasone 8 mg intravenously and, where necessary, salbutamol 200 mg to 300 mg intravenously or 500 mg intramuscularly.
Control
Participants in the control group received high‐flow oxygen treatment (8 L/min to10 L/min) administered by a non‐rebreather face mask and bronchodilators delivered by nebulization with oxygen at flows of 6 L/min to 8 L/min. Oximeters were used to measure oxygen saturation in those receiving high‐flow oxygen. All participants received other standard treatment according to Tasmanian Ambulance Service guidelines, including basic support, nebulised bronchodilators (salbutamol 5 mg made up in 2.5 mL normal saline, ipratropium bromide 500 μg made up with 2.5 mL normal saline), dexamethasone 8 mg intravenously and, where necessary, salbutamol 200 mg to 300 mg intravenously or 500 mg intramuscularly.
For more information about study characteristics, see Characteristics of included studies.
Excluded studies
Twenty studies were identified as excluded but relevant to the review (23 records). The primary reason for exclusion was the lack of randomisation of participants, found in 11 studies (Characteristics of excluded studies). Three of these studies were reviews, one a clinical audit and the remaining studies were not randomised. Four studies were not conducted in the pre‐hospital setting, but instead were undertaken once patients were admitted to hospital; one study did not have an oxygen comparator arm; and the remaining four studies were not conducted in patient cohorts with a majority diagnosis of AECOPD (60% or more).
Risk of bias in included studies
Risk of bias is reported for Austin 2010 and is displayed visually in Figure 3.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
Allocation
The method of sequence generation (computer random number generation after stratification by rurality) was assessed as being at low risk of bias. Allocation concealment was assessed as representing unclear risk of bias since the method of allocation was not reported; however, the study authors state that stratification by rurality was performed to reduce differences associated with transportation time between urban and rural areas, ensuring that treatment allocation was concealed before randomisation.
Blinding
The study authors report that paramedics, the research team and hospital staff were not blinded to treatment after randomisation. Therefore, we assessed the study as being at high risk of bias concerning blinding of participants. A respiratory physician blinded to treatment allocation reviewed lung function data and smoking history, from private and public medical records; however, no mention was made regarding the blinding for other outcome assessors so we assessed the study as being at unclear risk of detection bias.
Incomplete outcome data
Data were reported to be missing from both the ambulance and hospital patient records, resulting in denominators varying among analyses. Intention‐to‐treat analysis occurred, however, we assessed this domain as high risk of bias.
Selective reporting
No study protocol was available to determine whether selective reporting occurred, therefore we assessed the study as being at unclear risk of bias for this domain.
Other potential sources of bias
No other biases were identified.
Effects of interventions
High flow versus titrated oxygen therapy
All data below are reported for the one included study (Austin 2010).
Primary outcome
Mortality (respiratory‐related and all‐cause)
Among the cohort with confirmed AECOPD, a significant difference in pre/in‐hospital mortality was observed; there were 11 deaths out of 117 participants receiving high‐flow oxygen, compared to two deaths out of 97 participants receiving titrated oxygen (RR 0.22, 95% CI 0.05 to 0.97; 214 participants, 1 study; Analysis 1.1). This translates to a number needed to treat for an additional beneficial outcome (NNTB) of 14 (95% CI 12 to 355) with administration of titrated oxygen therapy, and is shown as a Cates plot in Figure 1. All deaths occurred after arrival at the hospital, including two in the emergency department and two in intensive care. As reported in the paper, the cause of mortality was respiratory failure in all cases, with approximately 70% of deaths occurring within the first five days following admission for both treatment arms.
Secondary outcomes
Quality of life
Quality of life was not reported as an outcome in this study.
Arterial blood gas
Based on the intention‐to‐treat analysis for the AECOPD subgroup, the difference between treatment arms for blood gas (pH) measurements observed between groups was uncertain (MD 0.06, 95% CI ‐0.04 to 0.16; 38 participants, 1 study; Analysis 1.2). Given that arterial blood gas was performed within 30 minutes of hospital arrival for only 11% of participants, it is important to note outcomes of the per‐protocol analysis. The per‐protocol analysis reported in the paper indicates significantly less respiratory acidosis (P = 0.01) and acute hypercapnia (P = 0.02) among participants receiving titrated oxygen (intervention) compared to those receiving high‐flow oxygen (control).
Lung function
Lung function was not reported as an outcome in this study.
Dyspnoea score
Dyspnoea was not reported as an outcome in this study.
Treatment failure requiring intensive care unit (ICU), invasive or non‐invasive ventilation
The difference observed between treatment arms for ventilation of any type in the intention‐to‐treat analysis was uncertain (RR 0.67, 95% CI 0.30 to 1.50; 189 participants, 1 study; Analysis 1.3); the within‐study subgroup analysis between invasive and non‐invasive ventilation is available in Analysis 2.1, however, this is not adjusted for clustering.
Hospital utilisation (length of stay, readmission rate, emergency department presentations)
The difference observed between treatment arms in length of hospital stay for the intention‐to‐treat analysis was also uncertain (MD ‐0.88 days, 95% CI ‐2.25 to 0.49; 214 participants, 1 study; Analysis 1.4). Other types of hospital utilisation were not reported as outcomes in this study.
Discussion
Summary of main results
This review update identified one new study that satisfied the pre‐defined inclusion criteria (Austin 2010). The study compared high‐flow oxygen therapy to a titrated approach in people with AECOPD in the pre‐hospital setting. Data from a subset of participants from Austin 2010 with a confirmed diagnosis of AECOPD were included for review. Two studies identified as ongoing in the original review were marked as awaiting classification (Eiser 2004; Elliott 2004) as there were no data available for analysis and attempts to contact the authors were unsuccessful. The included study was a well‐reported, well‐conducted Australian cluster‐RCT (Austin 2010).
For the primary outcome of mortality (respiratory‐related and all‐cause) there was a higher risk of death (9%; 11 individuals) with a high‐flow treatment approach compared to a titrated oxygen approach (2%; 2 individuals). All deaths were reportedly caused by respiratory failure and occurred once the patient arrived at hospital; 15% of reported deaths occurred in the emergency department and 15% occurred in the intensive care unit. With regard to arterial blood gas, Austin 2010 reported no evidence of an effect with high‐flow compared to titrated oxygen therapy in the intention‐to‐treat analysis; however, participants who were treated per protocol with a titrated oxygen therapy approach were less likely to have respiratory acidosis due to acute hypercapnia. There was no evidence of a difference between high‐flow and titrated oxygen therapy for the outcomes treatment failure requiring invasive or non‐invasive ventilation, or hospital utilisation.
Overall completeness and applicability of evidence
The overall completeness and applicability of evidence was low given that only one study was identified for inclusion in this review, covering only one potential comparison (high‐flow versus titrated oxygen therapy) with a total of 214 participants with AECOPD. Furthermore, this study was conducted in a single metropolitan, high‐income setting with older participants (with a mean age of approximately 68 years) who had access to an ambulance service and as such generalisability of the results to other contexts may be limited.
The included study reported the primary outcome pre‐specified in our review (mortality (respiratory‐related and all‐cause)) and some of our secondary outcomes (arterial blood gas, hospital utilisation and treatment failure); however, several important secondary outcomes were not measured (e.g. quality of life, lung function and dyspnoea). While perhaps difficult to measure in a pre‐hospital/emergency setting, these outcomes are nevertheless clinically relevant and important when considering the impact of this intervention on the patient's burden of disease. Further, protocol violation is a concern in the included study; only 11% of participants had their arterial blood gas collected per protocol and over half of the titrated oxygen group received inappropriate high‐flow oxygen therapy during the study event. High levels of protocol violation are undesirable with the potential to confound the interpretation of results. Arguably in this instance erroneous administration of high‐flow oxygen to the titrated oxygen group would have lessened the treatment effect, however, a significant reduction in pre/in‐hospital mortality was still detected in favour of the titrated oxygen group.
No studies were identified that compared high‐flow oxygen to placebo or standard care, though this is not surprising considering the ethical considerations of withholding first‐line treatment from a participant suffering with AECOPD. No studies were found evaluating the effect of low‐flow oxygen therapy. The findings of this review are therefore limited by a paucity of available data and an inability to assess all pre‐defined outcomes.
Certainty of the evidence
The certainty of the evidence was low. Certainty was downgraded in the domain of indirectness, because of the inclusion of only one study that included participants from only one location and health service. We further downgraded the certainty of the evidence for imprecision, because the low number of events resulted in wide confidence intervals with the upper limits approaching or including no difference. Other domains assessed, including risk of bias, inconsistency and publication bias, presented no cause for us to downgrade our assessment of certainty. Furthermore, the moderate sample size (214 participants) and methodologically rigorous study design does facilitate the ability to draw some conclusions for clinical practice in the absence of other available data.
Potential biases in the review process
Every effort was made to reduce the impact of bias on this review. A pre‐specified methodology was adhered to based on Cochrane standards (Higgins 2011). The systematic search for this review was conducted through diverse means (i.e. electronic databases, conference proceedings, author contact and online clinical trial registries) by an experienced Information Specialist and experienced review team in order to identify all available and potentially relevant studies. However, considering the lack of available evidence returned by this search, it is likely that some publication bias was introduced leading to over‐ or under‐estimation of the intervention effects, or indeed the inability to report on some outcomes altogether. From the original publication of this review, two studies have been moved from ongoing to awaiting classification (Eiser 2004; Elliott 2004). This is likely illustrative of the difficulty authors face in having their results published and the ultimate lack of evidence available for inclusion in this review. Attempts were made to overcome this by contacting the authors or listed contact persons (or both) for these studies; however, no responses were received.
While there may have been potential for false exclusion of relevant studies and data entry error, steps were taken to mitigate this by having two independent authors screen, extract and check data entry performed. The only included study was written by the two original authors of this review (MA and RW). The publication of Austin 2010 occurred subsequent to this review. As a result, this may have been a source of bias, however, to mitigate this issue MA and RW were not involved in any capacity for protocol revision, study screening and selection, data extraction and analysis of the included and excluded studies.
Agreements and disagreements with other studies or reviews
The traditional approach to treatment of patients with shortness of breath is high‐flow oxygen adopted on the basis that the population as a whole are at greater risk from failure to correct oxygenation than from its excessive administration (Kane 2013). The results of this review somewhat support a retrospective study (Denniston 2002) that found that the administration of an inspired oxygen concentration above 28% was associated with a higher in‐hospital mortality (of 14%) than administration of a concentration of oxygen less that 28% (mortality of 2%). Though Denniston 2002 did not directly assess titrated oxygen therapy, it suggested that the continued use of a high‐flow approach was no longer appropriate. However, the non‐randomised design meant that the results could have been confounded by the severity of illness influencing the choice of oxygen concentration used. The studies identified in this review as awaiting classification have, as with many pre‐hospital studies, experienced difficulties with design, implementation and compliance of staff. This is likely a result of entrenched habits of treating paramedics. Future studies attempting a similar undertaking may consider engaging organisational executives in endorsing the study and its methods, to encourage adoption of the protocol and overcome reservations about change of practice to an experimental treatment option. Further, measurements may be best performed by a researcher rather than left to clinical staff to ensure quality data and results. This observation, however, should not prevent researchers, health professionals and policy makers from striving to obtain evidence to identify the optimal oxygen treatment for AECOPD.
Recently, guideline recommendations have moved away from high‐flow oxygen therapy in AECOPD in general, towards a more titrated approach (Beasley 2015; Kane 2013; O'Driscoll 2017). It appears that there is a current tendency toward risk‐adverse practice culture as this recommendation is presently supported by only single, predominantly non‐randomised, studies; our review (which identified only one RCT) only serves to support this. Given the included RCT was published in 2010, and recent literature searches uncover no new studies, it is perhaps reasonable to presume that given the stakes the existing evidence for a titrated approach to oxygen delivery is considered widely to be 'good enough' in the absence of a clear demonstrable benefit to administration of high‐flow oxygen in this population.

In the high‐dose oxygen group 9 people out of 100 died, compared to 2 (95% confidence interval 0 to 9) out of 100 for the titrated oxygen group.

Study flow diagram.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.

Comparison 1 High flow versus titrated oxygen therapy, Outcome 1 Mortality (respiratory‐related and all‐cause).

Comparison 1 High flow versus titrated oxygen therapy, Outcome 2 Arterial Blood Gas (pH).

Comparison 1 High flow versus titrated oxygen therapy, Outcome 3 Ventilation of any type.

Comparison 1 High flow versus titrated oxygen therapy, Outcome 4 Length of Stay (days).
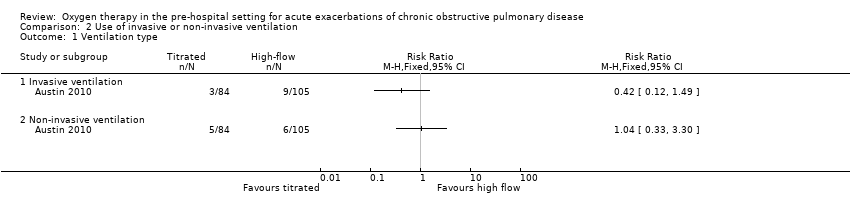
Comparison 2 Use of invasive or non‐invasive ventilation, Outcome 1 Ventilation type.
| Titrated oxygen therapy compared to high‐flow oxygen therapy for acute exacerbations of chronic obstructive pulmonary disease | ||||||
| Patient or population: adults with acute exacerbations of chronic obstructive pulmonary disease | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | № of participants | Certainty of the evidence | Comments | |
| Risk with high‐flow oxygen therapy | Risk with titrated oxygen therapy | |||||
| Mortality (respiratory‐ related and all‐cause | 94 per 1,000 | 21 per 1,000 | RR 0.22 | 214 | ⊕⊕⊝⊝ | A difference in mortality was observed, with 11 deaths in the high‐flow oxygen arm compared to two deaths in the titrated oxygen arm (P = 0.05). This translates to a number needed to treat for an additional beneficial outcome (NNTB) of 14 (95% CI 12 to 355) with administration of titrated oxygen therapy, and is shown as a Cates plot in Figure 1. All deaths occurred after arrival at the hospital; two were in intensive care. Respiratory failure was the cause of mortality in all cases, with approximately 70% of deaths occurring within the first five days following admission for both treatment arms. |
| Arterial blood gas (pH) | The mean arterial blood gas (pH) was 7.29 | MD 0.06 pH higher | ‐ | 214 | ⊕⊕⊝⊝ | Based on the intention‐to‐treat analysis for the COPD subgroup, no significant difference between treatment arms for blood gas measurements was observed between groups (P = 0.23). Only 11% of participants had this measurement performed according to protocol. |
| Ventilation of any type | 143 per 1,000 | 96 per 1000 | RR 0.67 | 189 | ⊕⊕⊝⊝ | No significant difference observed between treatment arms for ventilation requirement for per protocol or intention‐to‐treat analyses. |
| Length of hospital stay | The mean length of hospital stay was 6.3 days | MD 0.88 days lower | ‐ | 214 | ⊕⊕⊝⊝ | No significant difference was observed between treatment arms in length of hospital stay for the intention‐to‐treat analysis (P = 0.21). |
| Quality of life ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| Lung function ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| Dyspnoea score ‐ not measured | ‐ | ‐ | ‐ | ‐ | ‐ | This was not reported as an outcome in the single included study. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1 Downgraded once for indirectness because there was a single included study with moderate sample size (n = 214). The study was conducted in one state within Australia only. | ||||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Mortality (respiratory‐related and all‐cause) Show forest plot | 1 | Risk Ratio (Fixed, 95% CI) | Totals not selected | |
| 2 Arterial Blood Gas (pH) Show forest plot | 1 | Mean Difference (Fixed, 95% CI) | Totals not selected | |
| 3 Ventilation of any type Show forest plot | 1 | Risk Ratio (Fixed, 95% CI) | Totals not selected | |
| 4 Length of Stay (days) Show forest plot | 1 | Mean Difference (Fixed, 95% CI) | Totals not selected | |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Ventilation type Show forest plot | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.1 Invasive ventilation | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 1.2 Non‐invasive ventilation | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |

