Sedutan kortikosteroid lewat (≥ 7 hari) untuk mengurangkan displasia bronkopulmonary dalam bayi pramatang
Appendices
Appendix 1. Standard search methodology
PubMed: ((infant, newborn[MeSH] OR newborn OR neonate OR neonatal OR premature OR low birth weight OR VLBW OR LBW or infan* or neonat*) AND (randomized controlled trial [pt] OR controlled clinical trial [pt] OR randomized [tiab] OR placebo [tiab] OR drug therapy [sh] OR randomly [tiab] OR trial [tiab] OR groups [tiab]) NOT (animals [mh] NOT humans [mh]))
Embase: (infant, newborn or newborn or neonate or neonatal or premature or very low birth weight or low birth weight or VLBW or LBW or Newborn or infan* or neonat*) AND (human not animal) AND (randomized controlled trial or controlled clinical trial or randomized or placebo or clinical trials as topic or randomly or trial or clinical trial)
CINAHL: (infant, newborn OR newborn OR neonate OR neonatal OR premature OR low birth weight OR VLBW OR LBW or Newborn or infan* or neonat*) AND (randomized controlled trial OR controlled clinical trial OR randomized OR placebo OR clinical trials as topic OR randomly OR trial OR PT clinical trial)
Cochrane Library: (infant or newborn or neonate or neonatal or premature or preterm or very low birth weight or low birth weight or VLBW or LBW)
Appendix 2. Previous search methodology
Previous versions of this review used the following Medical Subject Heading terms (MeSH) and text words:
(steroids or glucocorticoids or flixotide or fluticasone or becotide or beclomethasone or pulmicort or budesonide or anti inflammatory agents) and (inhal* or nebulis* or nebuliz* or aerolis* or aeroliz*) and (neonatal chronic lung disease or bronchopulmonary dysplasia or neonatal respiratory distress syndrome or chronic lung disease of prematurity or chronic lung disease of infancy).
No search limits were used.
Appendix 3. 'Risk of bias' tool
We used the standard methods of Cochrane and Cochrane Neonatal to assess the methodological quality (to meet the validity criteria) of the trials. For each trial, we sought information regarding the method of randomisation and the blinding and reporting of all outcomes of all the infants enrolled in the trial. We assessed each criterion as low, high, or unclear risk. Two review authors separately assessed each study. Any disagreements were resolved by discussion. We added this information to the Characteristics of included studies table. We evaluated the following issues and entered the findings into the 'Risk of bias' table.
Sequence generation (checking for possible selection bias). Was the allocation sequence adequately generated?
For each included study, we categorised the method used to generate the allocation sequence as:
-
low risk (any truly random process, e.g. random number table; computer random number generator);
-
high risk (any non‐random process, e.g. odd or even date of birth; hospital or clinic record number);
-
unclear risk.
Allocation concealment (checking for possible selection bias). Was allocation adequately concealed?
For each included study, we categorised the method used to conceal the allocation sequence as:
-
low risk (e.g. telephone or central randomisation; consecutively numbered, sealed, opaque envelopes);
-
high risk (open random allocation; unsealed or non‐opaque envelopes, alternation; date of birth);
-
unclear risk.
Blinding of participants and personnel (checking for possible performance bias). Was knowledge of the allocated intervention adequately prevented during the study?
For each included study, we categorised the methods used to blind study participants and personnel from knowledge of which intervention a participant received. Blinding was assessed separately for different outcomes or class of outcomes. We categorised the methods as:
-
low risk, high risk, or unclear risk for participants;
-
low risk, high risk, or unclear risk for personnel.
Blinding of outcome assessment (checking for possible detection bias). Was knowledge of the allocated intervention adequately prevented at the time of outcome assessment?
For each included study, we categorised the methods used to blind outcome assessment. Blinding was assessed separately for different outcomes or class of outcomes. We categorised the methods as:
-
low risk for outcome assessors;
-
high risk for outcome assessors;
-
unclear risk for outcome assessors.
Incomplete outcome data (checking for possible attrition bias through withdrawals, dropouts, protocol deviations). Were incomplete outcome data adequately addressed?
For each included study and for each outcome, we described the completeness of data including attrition and exclusions from the analysis. We noted whether attrition and exclusions were reported, the numbers included in the analysis at each stage (compared with the total randomised participants), reasons for attrition or exclusion where reported, and whether missing data were balanced across groups or were related to outcomes. Where sufficient information was reported or supplied by the trial authors, we re‐included missing data in the analyses. We categorised the methods as:
-
low risk (< 20% missing data);
-
high risk (≥ 20% missing data);
-
unclear risk.
Selective reporting bias. Are reports of the study free of the suggestion of selective outcome reporting?
For each included study, we described how we investigated the possibility of selective outcome reporting bias and what we found. We assessed the methods as:
-
low risk (where it is clear that all of the study's prespecified outcomes and all expected outcomes of interest to the review have been reported);
-
high risk (where not all the study's prespecified outcomes have been reported; one or more reported primary outcomes were not prespecified outcomes of interest and are reported incompletely and so cannot be used; study fails to include results of a key outcome that would have been expected to have been reported);
-
unclear risk.
Other sources of bias. Was the study apparently free of other problems that could put it at high risk of bias?
For each included study, we described any important concerns we had about other possible sources of bias (e.g. whether there was a potential source of bias related to the specific study design or whether the trial was stopped early due to some data‐dependent process). We assessed whether each study was free of other problems that could put it at risk of bias as:
-
low risk;
-
high risk;
-
unclear risk.
If needed, we explored the impact of the level of bias through the undertaking of sensitivity analyses.

Flow of inclusion of randomised controlled trials in different phases of search.

Study flow diagram: review update.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
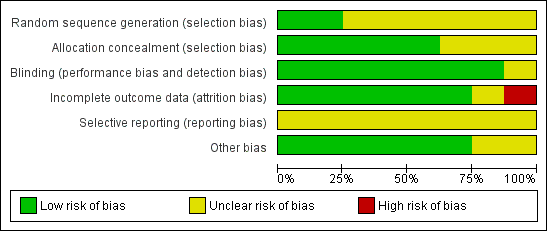
Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.
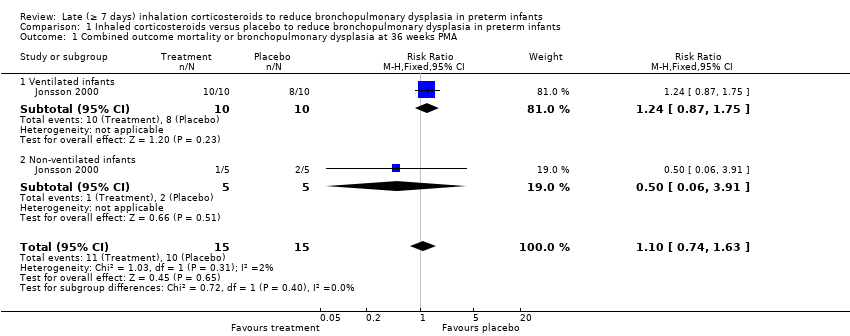
Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 1 Combined outcome mortality or bronchopulmonary dysplasia at 36 weeks PMA.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 2 Mortality at 28 days PNA.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 3 Mortality at 36 weeks PMA.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 4 Mortality at hospital discharge.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 5 Bronchopulmonary dysplasia at 28 days PNA.
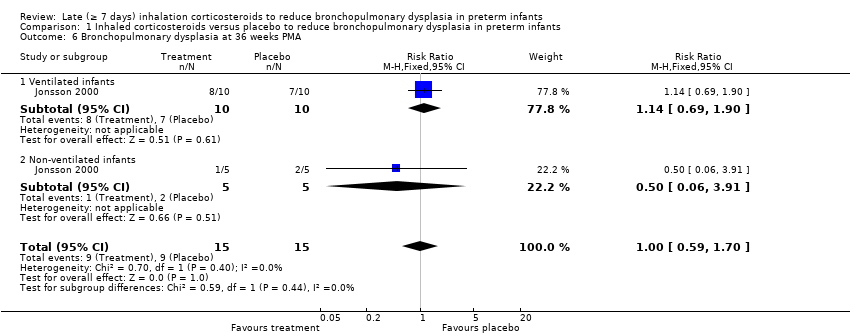
Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 6 Bronchopulmonary dysplasia at 36 weeks PMA.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 7 Combined outcome mortality and bronchopulmonary dysplasia at 28 days PNA.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 8 Failure to extubate day 7.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 9 Failure to extubate day 14.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 10 Failure to extubate at the latest reported moment.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 11 Days of mechanical ventilation.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 12 Days of supplemental oxygen.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 13 Open‐label intravenous corticosteroids.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 14 Sepsis (clinical suspected or culture proven).

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 15 Patent ductus arteriosus.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 16 Hypertension (> 2 SD).

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 17 Necrotising enterocolitis.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 18 Intraventricular haemorrhage (any grade).

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 19 Days of hospitalisation.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 20 Airway resistance.

Comparison 1 Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants, Outcome 21 Dynamic lung compliance.
| Inhaled corticosteroids versus placebo to reduce bronchopulmonary dysplasia in preterm infants | ||||||
| Patient or population: preterm infants | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect | № of participants | Quality of the evidence | Comments | |
| Risk with placebo | Risk with inhaled corticosteroids | |||||
| Combined outcome mortality or bronchopulmonary dysplasia at 36 weeks postmenstrual age | Study population | RR 1.10 | 30 | ⊕⊝⊝⊝ | ||
| 533 per 1000 | 587 per 1000 | |||||
| Mortality at 36 weeks postmenstrual age | Study population | RR 3.00 | 61 | ⊕⊕⊝⊝ | ||
| 0 per 1000 | 0 per 1000 | |||||
| Bronchopulmonary dysplasia at 36 weeks postmenstrual age | Study population | RR 1.00 | 30 | ⊕⊝⊝⊝ | ||
| 600 per 1000 | 600 per 1000 | |||||
| Open‐label intravenous corticosteroids | Study population | RR 0.51 | 74 | ⊕⊝⊝⊝ | ||
| 432 per 1000 | 320 per 1000 | |||||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: confidence interval; RCT: randomised controlled trial; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence | ||||||
| 1Risk of bias: No serious limitations. No downgrade. | ||||||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Combined outcome mortality or bronchopulmonary dysplasia at 36 weeks PMA Show forest plot | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.74, 1.63] |
| 1.1 Ventilated infants | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.24 [0.87, 1.75] |
| 1.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.06, 3.91] |
| 2 Mortality at 28 days PNA Show forest plot | 2 | 51 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.14, 65.90] |
| 2.1 Ventilated infants | 2 | 41 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.14, 65.90] |
| 2.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 3 Mortality at 36 weeks PMA Show forest plot | 3 | 61 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.35, 25.78] |
| 3.1 Ventilated infants | 3 | 51 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.35, 25.78] |
| 3.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 4 Mortality at hospital discharge Show forest plot | 3 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.35, 25.78] |
| 4.1 Ventilated infants | 3 | 43 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.35, 25.78] |
| 4.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5 Bronchopulmonary dysplasia at 28 days PNA Show forest plot | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.72, 1.21] |
| 5.1 Ventilated infants | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.61, 1.29] |
| 5.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.71, 1.41] |
| 6 Bronchopulmonary dysplasia at 36 weeks PMA Show forest plot | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.59, 1.70] |
| 6.1 Ventilated infants | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.14 [0.69, 1.90] |
| 6.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.06, 3.91] |
| 7 Combined outcome mortality and bronchopulmonary dysplasia at 28 days PNA Show forest plot | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.85, 1.18] |
| 7.1 Ventilated infants | 1 | 20 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.83, 1.20] |
| 7.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.71, 1.41] |
| 8 Failure to extubate day 7 Show forest plot | 5 | 79 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.66, 0.98] |
| 9 Failure to extubate day 14 Show forest plot | 2 | 27 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.36 [0.10, 1.33] |
| 10 Failure to extubate at the latest reported moment Show forest plot | 6 | 90 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.60 [0.45, 0.80] |
| 11 Days of mechanical ventilation Show forest plot | 3 | 45 | Mean Difference (IV, Fixed, 95% CI) | 3.42 [‐1.30, 8.13] |
| 12 Days of supplemental oxygen Show forest plot | 4 | 141 | Mean Difference (IV, Fixed, 95% CI) | 0.57 [‐5.92, 7.07] |
| 12.1 Ventilated infants | 4 | 100 | Mean Difference (IV, Fixed, 95% CI) | 5.53 [‐3.99, 15.05] |
| 12.2 Non‐ventilated infants | 2 | 41 | Mean Difference (IV, Fixed, 95% CI) | ‐3.74 [‐12.63, 5.14] |
| 13 Open‐label intravenous corticosteroids Show forest plot | 4 | 74 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.26, 1.00] |
| 13.1 Ventilated infants | 4 | 64 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.26, 1.00] |
| 13.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 14 Sepsis (clinical suspected or culture proven) Show forest plot | 5 | 107 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.50, 1.64] |
| 14.1 Ventilated infants | 5 | 97 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.44, 1.77] |
| 14.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.36, 2.75] |
| 15 Patent ductus arteriosus Show forest plot | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.16, 6.20] |
| 15.1 Ventilated infants | 1 | 30 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.16, 6.20] |
| 16 Hypertension (> 2 SD) Show forest plot | 1 | 27 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16.1 Ventilated infants | 1 | 17 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 16.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17 Necrotising enterocolitis Show forest plot | 1 | 27 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.1 Ventilated infants | 1 | 17 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 17.2 Non‐ventilated infants | 1 | 10 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 18 Intraventricular haemorrhage (any grade) Show forest plot | 1 | 19 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.6 [0.13, 2.82] |
| 18.1 Ventilated infants | 1 | 19 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.6 [0.13, 2.82] |
| 19 Days of hospitalisation Show forest plot | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐24.70 [‐41.75, ‐7.65] |
| 19.1 Ventilated infants | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐24.70 [‐41.75, ‐7.65] |
| 20 Airway resistance Show forest plot | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | 21.40 [‐71.11, 113.91] |
| 21 Dynamic lung compliance Show forest plot | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐0.22 [‐0.33, ‐0.11] |

