Contenido relacionado
Revisiones y protocolos relacionados
Sherab Bhutia, Peng F Wong | 16 julio 2013
Lindsay Robertson, Lauren E Jones | 9 febrero 2017
Aniek AG Zee, Kelly van Lieshout, Maaike van der Heide, Loes Janssen, Heinrich MJ Janzing | 6 agosto 2017
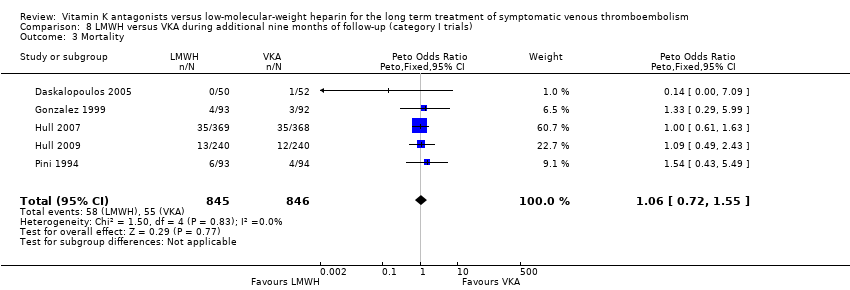
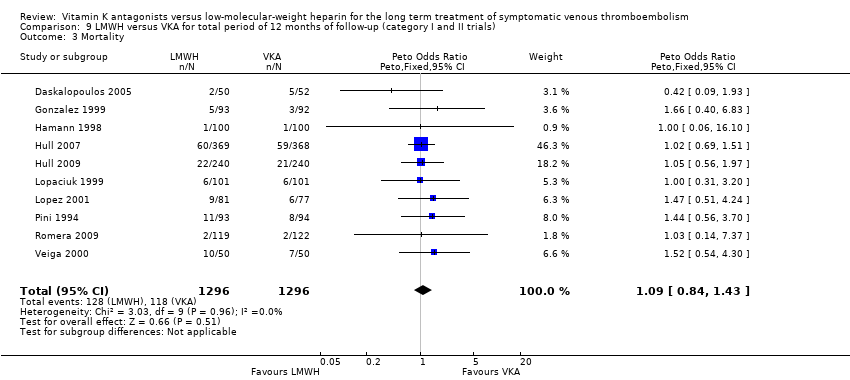
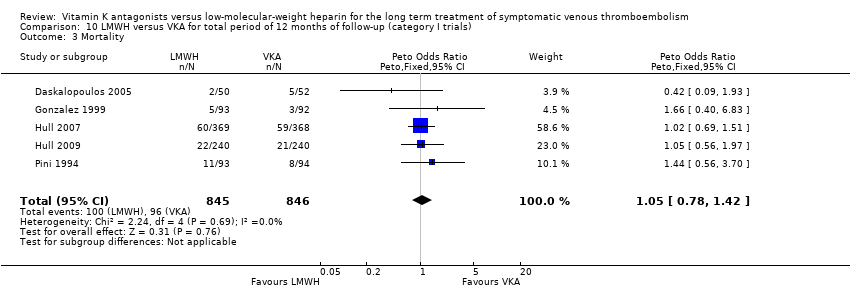
Saskia Middeldorp, Martin H Prins, Barbara A Hutten | 5 agosto 2014
Carlos A Salazar, German Malaga, Giuliana Malasquez | 14 abril 2010
Lindsay Robertson, James Strachan | 14 febrero 2017
Marie-Claude Pelland-Marcotte, Nour Amiri, Maria L Avila, Leonardo R Brandão | 18 junio 2020
Benilde Cosmi, Eleonora Conti, Sergio Coccheri | 7 mayo 2014
Anne WS Rutjes, Ettore Porreca, Matteo Candeloro, Emanuele Valeriani, Marcello Di Nisio | 18 diciembre 2020
Gustavo MS Brandao, Daniela R Junqueira, Hamilton A Rollo, Marcone L Sobreira | 2 diciembre 2017