Related content
Related reviews and protocols
Vivienne Kirchin, Tobias Page, Phil E Keegan, Kofi OM Atiemo, June D Cody, Samuel McClinton, Patricia Aluko | 25 July 2017
Dimitri Beeckman, Nele Van Damme, Lisette Schoonhoven, Aurélie Van Lancker, Jan Kottner, Hilde Beele, Mikel Gray, Sue Woodward, Mandy Fader, Karen Van den Bussche, Ann Van Hecke, Dorien De Meyer, Sofie Verhaeghe | 14 November 2016
Thomas BL Lam, Muhammad Imran Omar, Euan Fisher, Katie Gillies, Sara MacLennan | 23 September 2014
Laercio A Silva, Régis B Andriolo, Álvaro N Atallah, Edina MK da Silva | 27 September 2014
Mari Imamura, Kate Williams, Mandy Wells, Catherine McGrother | 2 December 2015
Muhammad Imran Omar, Cameron Edwin Alexander | 11 June 2013
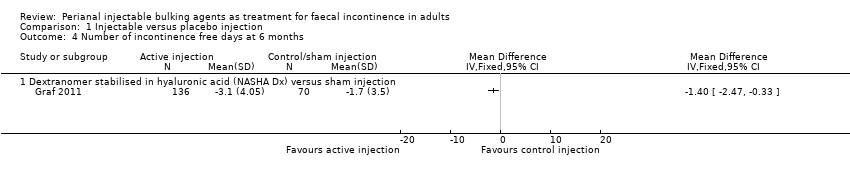
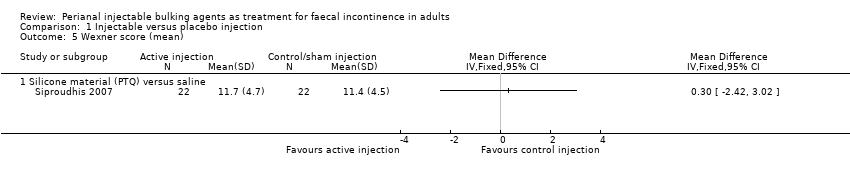
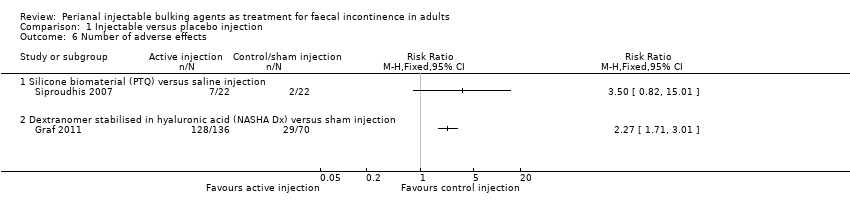
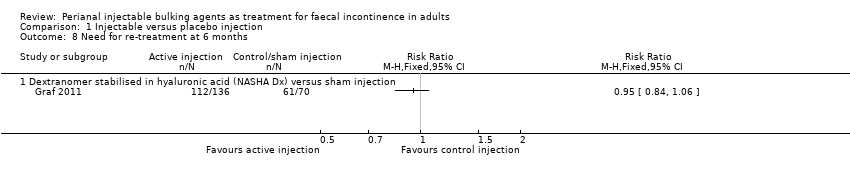
Mohamed A Thahaa, Amin A Abukara, Noel N Thin, Anthony Ramsanahie, Charles H Knowles | 24 August 2015
James B Duthie, Michael Vincent, G Peter Herbison, David Iain Wilson, Don Wilson | 7 December 2011
Gordon Hosker, June D Cody, Christine C Norton | 18 July 2007
Ashley J Shepherd, William G Mackay, Suzanne Hagen | 6 March 2017