Intranasal steroids for acute sinusitis
Information
- DOI:
- https://doi.org/10.1002/14651858.CD005149.pub4Copy DOI
- Database:
-
- Cochrane Database of Systematic Reviews
- Version published:
-
- 02 December 2013see what's new
- Type:
-
- Intervention
- Stage:
-
- Review
- Cochrane Editorial Group:
-
Cochrane Acute Respiratory Infections Group
- Copyright:
-
- Copyright © 2019 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Article metrics
Altmetric:
Cited by:
Authors
Contributions of authors
Dr Anca Zalmanovici wrote the review, searched the literature, selected the studies to be included, assessed their quality, entered the data into RevMan 2012, wrote the methods, results and discussion sections and updated the review.
Dr John Yaphe searched the literature, was an independent assessor in selecting trials to be included, assessed the quality of the trials, wrote the discussion section and edited the review.
Declarations of interest
None known.
Acknowledgements
Thanks to the Cochrane Acute Respiratory Infections Group for ongoing assistance, especially Chanpen Choprapawon, Leonard Leibovici, Marilyn Oates, Richmal Oates‐Whitehead and Erik Schenkel for useful comments on the draft protocol. We wish to thank Chanpen Choprapawon, Eric Schenkel, Richard G. Shoemaker and Leonard Leibovici for the commenting on the draft review and Liz Dooley and Sarah Thorning for the suggested search strategy and support with the searches and updates. We also wish to thank Yusra Adel Badr, JM Klossek, Sree Nair and Nick Matheson for commenting on the 2009 updated draft and Clalit Health Services Tel‐Aviv district for supporting the activities of Anca Zalmanovici.
Version history
| Published | Title | Stage | Authors | Version |
| 2013 Dec 02 | Intranasal steroids for acute sinusitis | Review | Anca Zalmanovici Trestioreanu, John Yaphe | |
| 2009 Oct 07 | Intranasal steroids for acute sinusitis | Review | Anca Zalmanovici Trestioreanu, John Yaphe | |
| 2009 Jul 08 | Steroids for acute sinusitis | Review | Anca Zalmanovici, John Yaphe | |
| 2005 Jan 24 | Steroids for acute sinusitis | Protocol | Anca Zalmanovici, John Yaphe | |
Notes
We thank Professor Leonard Leibovici from the Rabin Medical Center and Professor Michael A Weingarten, of the Department of Family Medicine, Tel‐Aviv University, for their useful suggestions and final revision of this review.
Keywords
MeSH
Medical Subject Headings (MeSH) Keywords
Medical Subject Headings Check Words
Adult; Child; Humans;
PICOs
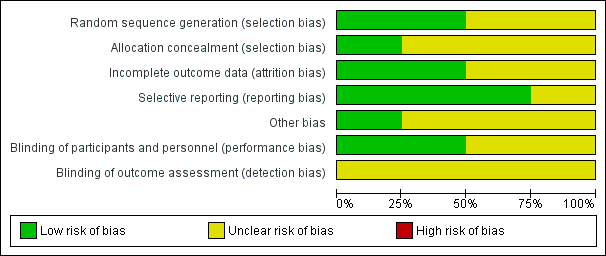
'Risk of bias' graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included study.
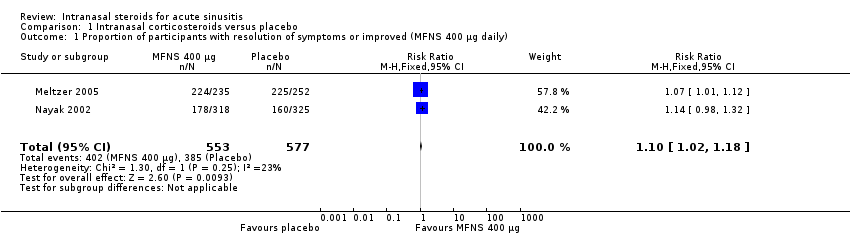
Comparison 1 Intranasal corticosteroids versus placebo, Outcome 1 Proportion of participants with resolution of symptoms or improved (MFNS 400 µg daily).

Comparison 1 Intranasal corticosteroids versus placebo, Outcome 2 Proportion of participants with resolution of symptoms or improved (MFNS 200 µg daily).
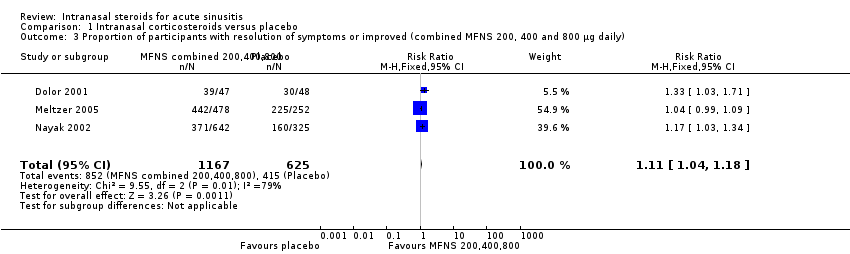
Comparison 1 Intranasal corticosteroids versus placebo, Outcome 3 Proportion of participants with resolution of symptoms or improved (combined MFNS 200, 400 and 800 µg daily).

Comparison 1 Intranasal corticosteroids versus placebo, Outcome 4 Number of participants that dropped out from the study (MFNS 400 µg daily).

Comparison 1 Intranasal corticosteroids versus placebo, Outcome 5 Number of participants that dropped out from the study (MFNS 200 µg daily).

Comparison 1 Intranasal corticosteroids versus placebo, Outcome 6 Number of participants that dropped out from the study (combined MFNS 200, 400 and 800 µg daily).

Comparison 1 Intranasal corticosteroids versus placebo, Outcome 7 Relapse (combined 200 and 400 µg daily).
| Study | Intervention | Side effects | Comments |
| Fluticasone propionate 2 puffs ‐ total dose 200 µg or placebo nasal spray once daily in addition to 250 mg cefuroxime axetil orally twice daily and 2 puffs of xylometazoline hydrochloride twice daily | Headache, bloody nose, vaginal itching, yeast infection, nausea, stomach irritation, diarrhoea, increased congestion, hay fever, light‐headed, sore throat, thirsty, itching, rash, cough, fatigue, metallic taste, felt dried out, nasal tissue felt inflamed | No serious unexpected adverse events reported | |
| Amoxicillin‐clavulanate potassium 875 mg | Epistaxis was the most frequently reported adverse event | Treatment well‐tolerated, adverse events similar for all 3 arms of mild/moderate intensity: 12%, 15%, 15% in the MFNS 400, 800 µg and placebo arms | |
| Budesonide 50 µg or placebo nasal spray to each nostril bid in addition to amoxicillin clavulanate potassium 40 mg/kg/day tid | Rash after 1 week attributed to the antibiotic in 1 subject that was switched to cefaclor | No specific adverse events related to the INCS use were reported | |
| MFNS 200 µg once daily or twice daily nasal spray | Headache and epistaxis were most common reported | Most adverse events were mild or moderate with a similar incidence among treatment groups: 36.2%, | |
| bid: twice daily | |||
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Proportion of participants with resolution of symptoms or improved (MFNS 400 µg daily) Show forest plot | 2 | 1130 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.10 [1.02, 1.18] |
| 2 Proportion of participants with resolution of symptoms or improved (MFNS 200 µg daily) Show forest plot | 2 | 590 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.98, 1.11] |
| 3 Proportion of participants with resolution of symptoms or improved (combined MFNS 200, 400 and 800 µg daily) Show forest plot | 3 | 1792 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.11 [1.04, 1.18] |
| 4 Number of participants that dropped out from the study (MFNS 400 µg daily) Show forest plot | 2 | 1130 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.86 [0.61, 1.20] |
| 5 Number of participants that dropped out from the study (MFNS 200 µg daily) Show forest plot | 2 | 590 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.46, 1.21] |
| 6 Number of participants that dropped out from the study (combined MFNS 200, 400 and 800 µg daily) Show forest plot | 3 | 1792 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.64, 1.12] |
| 7 Relapse (combined 200 and 400 µg daily) Show forest plot | 2 | 825 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.44, 1.15] |

