Moduladores de los receptores de progesterona para la endometriosis
Appendices
Appendix 1. Cochrane Gynaecology and Fertility Group specialised register search strategy
From inception to 28 November 2016
Procite platform
Keywords CONTAINS "endometriosis" or "Endometriosis‐Symptoms" or "pelvic pain" or "dyspareunia" or Title CONTAINS "endometriosis" or "Endometriosis‐Symptoms" or "pelvic pain" or "dyspareunia"
AND
Keywords CONTAINS "mifepristone"or"RU486"or"gestrinone"or"Onapristone" or "asoprisnil" or "CBD 2914" or "CDB‐2914" or "CDB‐4124" or "Ulipristal" or "ulipristal acetate" or "progestagen receptor levels" or" progesterone receptor agonist" or "Progesterone Receptor Modulator" or "selective progesterone receptor modulator" or Title CONTAINS "mifepristone"or"RU486"or"gestrinone"or"Onapristone" or "asoprisnil" or "CBD 2914" or "CDB‐2914" or "CDB‐4124" or "Ulipristal" or "ulipristal acetate" or "progestagen receptor levels" or" progesterone receptor agonist" or "Progesterone Receptor Modulator" or "selective progesterone receptor modulator" (44 hits)
Appendix 2. CENTRAL CRSO search strategy
Searched 28 November 2016
Web platform
#1 MESH DESCRIPTOR Endometriosis EXPLODE ALL TREES 513
#2 Endometrio*:TI,AB,KY 1289
#3 (pelvi* pain):TI,AB,KY 878
#4 #1 OR #2 OR #3 1934
#5 MESH DESCRIPTOR Mifepristone EXPLODE ALL TREES 376
#6 mifepristone:TI,AB,KY 714
#7 RU486:TI,AB,KY 47
#8 (progest* adj1 antagonist*):TI,AB,KY 68
#9 (progest* adj1 modulator*):TI,AB,KY 33
#10 (ru 486 or ru486):TI,AB,KY 143
#11 ru38486:TI,AB,KY 5
#12 ru‐38486:TI,AB,KY 1
#13 MESH DESCRIPTOR Gestrinone EXPLODE ALL TREES 29
#14 Gestrinone:TI,AB,KY 58
#15 Onapristone:TI,AB,KY 9
#16 Aglepristone:TI,AB,KY 2
#17 anti‐progestin*:TI,AB,KY 2
#18 antiprogestin*:TI,AB,KY 47
#19 Asoprisnil:TI,AB,KY 10
#20 J867:TI,AB,KY 1
#21 Telapristone:TI,AB,KY 1
#22 CDB‐2914:TI,AB,KY 9
#23 Ulipristal:TI,AB,KY 37
#24 (SPRM* or PRAs or PRA):TI,AB,KY 788
#25 MESH DESCRIPTOR Receptors, Progesterone EXPLODE ALL TREES WITH QUALIFIERS AD,AG,AI,ME,TU 159
#26 #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15 OR #16 OR #17 OR #18 OR #19 OR #20 OR #21 OR #22 OR #23 OR #24 OR #25 1782
#27 #4 AND #26 65
Appendix 3. MEDLINE search strategy
From 1946 to 28 November 2016
Ovid platform
1 exp Endometriosis/ (19876)
2 Endometri$.tw. (84071)
3 exp Dyspareunia/ (1809)
4 Dyspareunia.tw. (3194)
5 pelvi$ pain.tw. (7650)
6 or/1‐5 (95322)
7 exp Mifepristone/ (5981)
8 mifepristone.tw. (3252)
9 RU486.tw. (2312)
10 (progest$ adj1 antagonist$).tw. (512)
11 (progest$ adj1 modulator$).tw. (11)
12 ru 486.tw. (1780)
13 mifegyne.tw. (13)
14 ru38486.tw. (409)
15 ru‐38486.tw. (465)
16 mifeprex.tw. (12)
17 r38486.tw. (1)
18 exp Gestrinone/ (198)
19 Gestrinone.tw. (192)
20 Lilopristone.tw. (20)
21 Onapristone.tw. (187)
22 Aglepristone.tw. (82)
23 (dimetriose or dimetrose).tw. (1)
24 Nemestran.tw. (4)
25 anti‐progestin$.tw. (151)
26 antiprogestin$.tw. (935)
27 Asoprisnil.tw. (47)
28 J867.tw. (13)
29 Telapristone.tw. (9)
30 CDB‐2914.tw. (48)
31 CDB‐4124.tw. (19)
32 Proellex.tw. (8)
33 Ulipristal.tw. (254)
34 (SPRM$ or PRAs).tw. (340)
35 exp Receptors, Progesterone/me, tu [Metabolism, Therapeutic Use] (9329)
36 or/7‐35 (18327)
37 6 and 36 (2106)
38 randomized controlled trial.pt. (469810)
39 controlled clinical trial.pt. (95074)
40 randomized.ab. (404455)
41 randomised.ab. (81254)
42 placebo.tw. (197050)
43 clinical trials as topic.sh. (189502)
44 randomly.ab. (285441)
45 trial.ti. (178951)
46 (crossover or cross‐over or cross over).tw. (76010)
47 or/38‐46 (1208254)
48 exp animals/ not humans.sh. (4669479)
49 47 not 48 (1114154)
50 37 and 49 (212)
Appendix 4. Embase search strategy
From 1980 to 28 November 2016
Ovid platform
1 exp endometriosis/ (30876)
2 Endometri$.tw. (104349)
3 Dyspareunia.tw. (5475)
4 pelvi$ pain.tw. (11229)
5 or/1‐4 (120810)
6 exp mifepristone/ (11579)
7 mifepristone.tw. (3888)
8 RU486.tw. (2483)
9 (progest$ adj1 antagonist$).tw. (521)
10 exp antigestagen/ (12402)
11 (progest$ adj1 modulator$).tw. (18)
12 exp progesterone receptor modulator/ (586)
13 ru 486.tw. (4148)
14 mifegyne.tw. (181)
15 ru38486.tw. (406)
16 ru‐38486.tw. (913)
17 mifeprex.tw. (111)
18 zk 98296.tw. (3)
19 r38486.tw. (1)
20 exp GESTRINONE/ (621)
21 Gestrinone.tw. (204)
22 Lilopristone.tw. (22)
23 Onapristone.tw. (216)
24 Aglepristone.tw. (80)
25 (dimetriose or dimetrose).tw. (14)
26 Nemestran.tw. (11)
27 anti‐progestin$.tw. (178)
28 antiprogestin$.tw. (968)
29 Asoprisnil.tw. (63)
30 J867.tw. (15)
31 Telapristone.tw. (16)
32 CDB‐2914.tw. (106)
33 CDB‐4124.tw. (47)
34 Proellex.tw. (22)
35 Ulipristal.tw. (466)
36 (SPRM$ or PRAs).tw. (596)
37 exp progesterone receptor/dt, pd [Drug Therapy, Pharmacology] (65)
38 or/6‐37 (14686)
39 5 and 38 (1698)
40 Clinical Trial/ (995452)
41 Randomized Controlled Trial/ (461742)
42 exp randomization/ (83639)
43 Single Blind Procedure/ (27251)
44 Double Blind Procedure/ (136941)
45 Crossover Procedure/ (53825)
46 Placebo/ (321968)
47 Randomi?ed controlled trial$.tw. (149359)
48 Rct.tw. (22353)
49 random allocation.tw. (1629)
50 randomly.tw. (338581)
51 randomly allocated.tw. (26583)
52 allocated randomly.tw. (2208)
53 (allocated adj2 random).tw. (843)
54 Single blind$.tw. (18663)
55 Double blind$.tw. (172862)
56 ((treble or triple) adj blind$).tw. (647)
57 placebo$.tw. (247461)
58 prospective study/ (386445)
59 or/40‐58 (1967839)
60 case study/ (92866)
61 case report.tw. (323319)
62 abstract report/ or letter/ (986309)
63 or/60‐62 (1393358)
64 59 not 63 (1916879)
65 (exp animal/ or animal.hw. or nonhuman/) not (exp human/ or human cell/ or (human or humans).ti.) (5725014)
66 64 not 65 (1793800)
67 39 and 66 (434)
Appendix 5. PsycINFO search strategy
From 1806 to 28 November 2016
Ovid platform
1 exp Dyspareunia/ (240)
2 Dyspareunia.tw. (528)
3 Endometriosis.tw. (209)
4 pelvi$ pain.tw. (489)
5 or/1‐4 (1140)
6 mifepristone.tw. (226)
7 RU486.tw. (159)
8 ru 486.tw. (88)
9 Gestrinone.tw. (0)
10 anti‐progestin$.tw. (4)
11 antiprogestin$.tw. (18)
12 (progest$ adj1 antagonist$).tw. (19)
13 Ulipristal.tw. (3)
14 SPRM$.tw. (3)
15 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 (434)
16 5 and 15 (3)
17 random.tw. (48413)
18 control.tw. (375365)
19 double‐blind.tw. (20296)
20 clinical trials/ (10020)
21 placebo/ (4732)
22 exp Treatment/ (669135)
23 or/17‐22 (1033445)
24 16 and 23 (1)

Risk of bias graph: review authors' judgements about each risk of bias item presented as percentages across all included studies.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study.
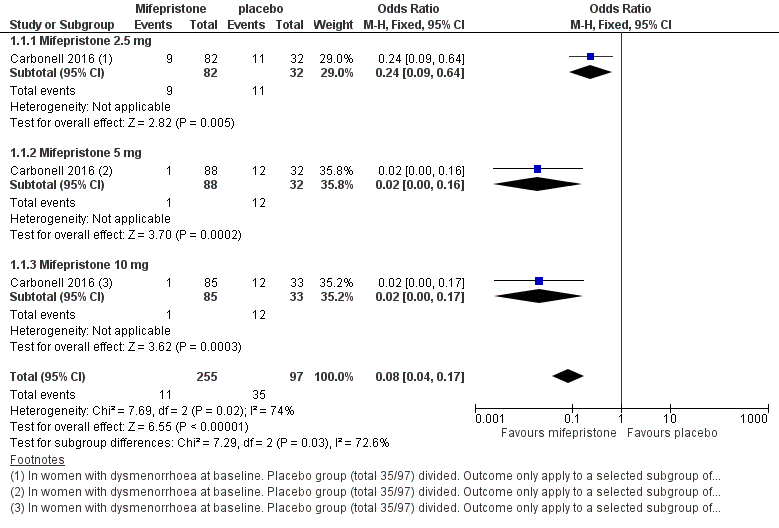
Forest plot of comparison: 1 Effectiveness of mifepristone versus placebo, patient‐assessed outcomes, outcome: 1.1 Dysmenorrhoea at three months.

Forest plot of comparison.
2 Mifepristone versus placebo, patient‐assessed outcomes, outcome: 2.1 Amenorrhoea at three months.

Forest plot of comparison: side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, outcome: 2.2 Hot flushes at three months.
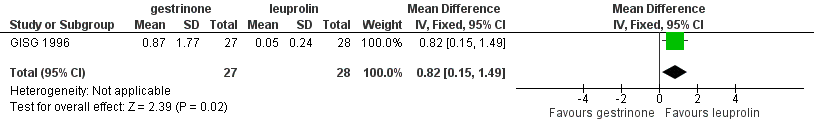
Forest plot of comparison: 7 Gestrinone versus leuprolin for six months: efficacy and side effects, outcome: 7.1 Painful periods, visual analogue scale.

Forest plot of comparison: 7 Gestrinone versus leuprolin for six months: efficacy and side effects, outcome: 7.3 Pain on intercourse, visual analogue scale.

Comparison 1 Effectiveness of mifepristone versus placebo, patient‐assessed outcomes, Outcome 1 Dysmenorrhoea at 3 months.

Comparison 1 Effectiveness of mifepristone versus placebo, patient‐assessed outcomes, Outcome 2 Dyspareunia at 3 months.

Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 1 Amenorrhoea at 3 months.
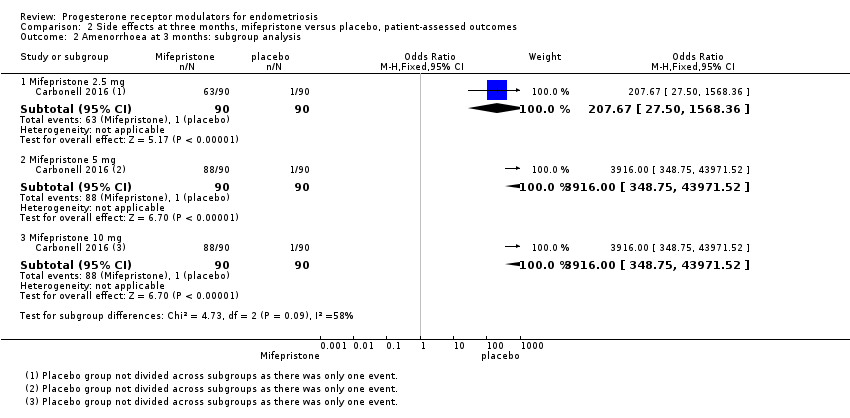
Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 2 Amenorrhoea at 3 months: subgroup analysis.

Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 3 Hot flushes at 3 months.

Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 4 Hot flushes at 3 months: subgroup analysis.
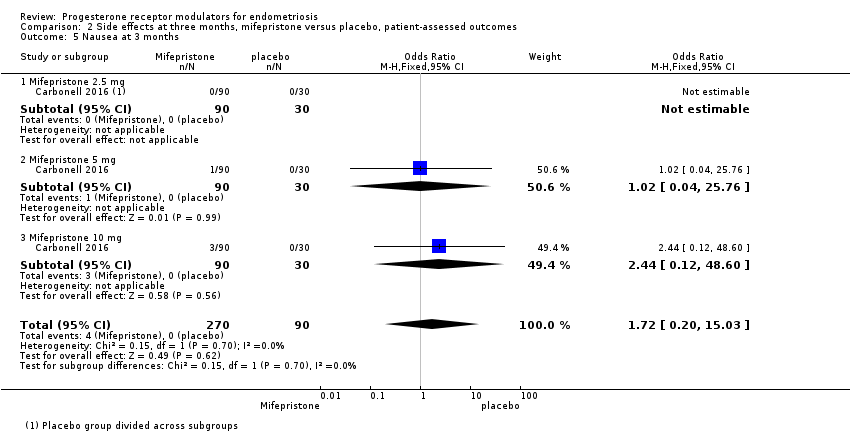
Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 5 Nausea at 3 months.
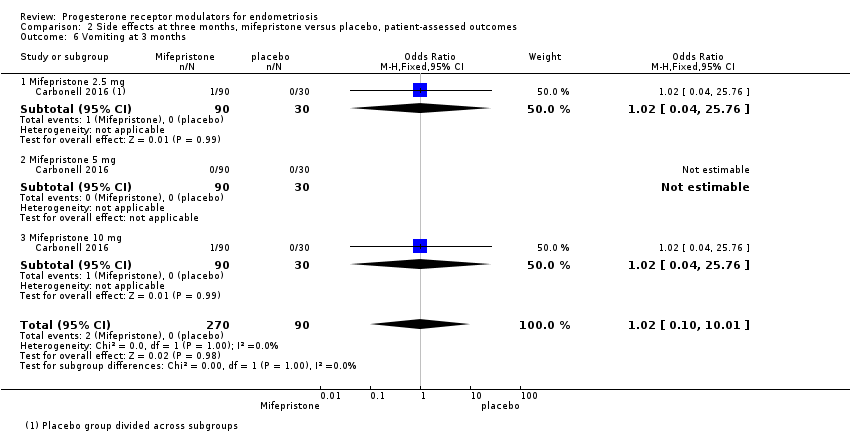
Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 6 Vomiting at 3 months.

Comparison 2 Side effects at three months, mifepristone versus placebo, patient‐assessed outcomes, Outcome 7 Fatigue/Tiredness at 3 months.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 1 Prevalence of dysmenorrhoea.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 2 Dysmenorrhoea score 0‐10 VAS scale.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 3 Prevalence of dyspareunia.
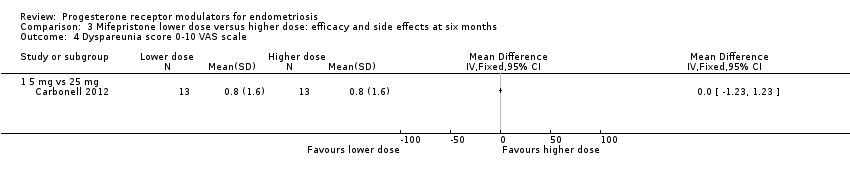
Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 4 Dyspareunia score 0‐10 VAS scale.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 5 Prevalence of pelvic pain.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 6 Prevalence of amenorrhoea.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 7 Prevalence of hot flushes.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 8 Prevalence of nausea.
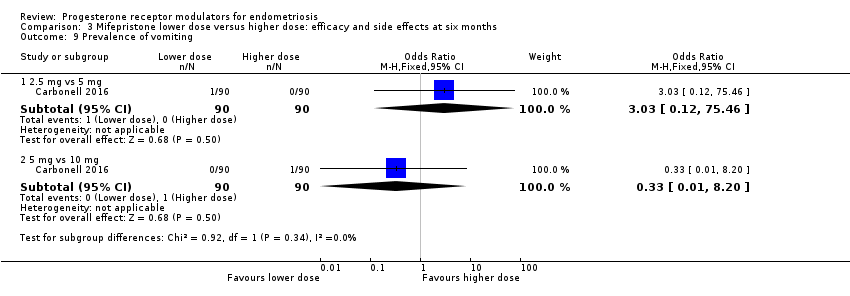
Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 9 Prevalence of vomiting.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 10 Prevalence of fatigue/tiredness.

Comparison 3 Mifepristone lower dose versus higher dose: efficacy and side effects at six months, Outcome 11 Prevalence of endometrial thickness > 20 mm.

Comparison 4 Gestrinone versus danazol for six months: efficacy and side effects, Outcome 1 None or mild pelvic pain.
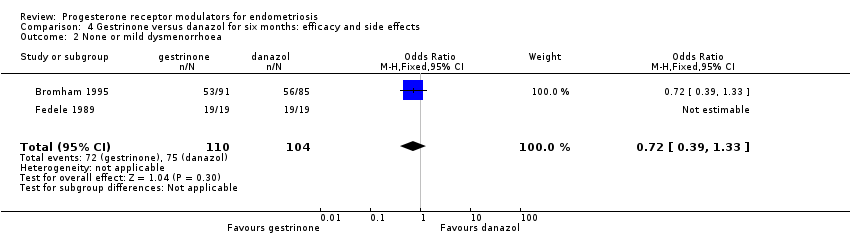
Comparison 4 Gestrinone versus danazol for six months: efficacy and side effects, Outcome 2 None or mild dysmenorrhoea.

Comparison 4 Gestrinone versus danazol for six months: efficacy and side effects, Outcome 3 None or mild dyspareunia.

Comparison 4 Gestrinone versus danazol for six months: efficacy and side effects, Outcome 4 Adverse effects.
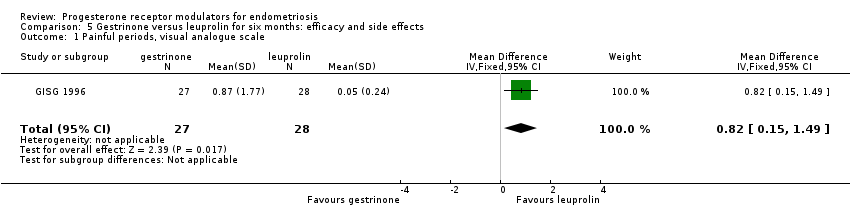
Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 1 Painful periods, visual analogue scale.
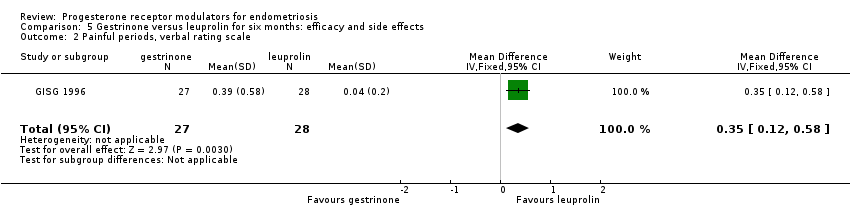
Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 2 Painful periods, verbal rating scale.

Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 3 Pain on intercourse, visual analogue scale.
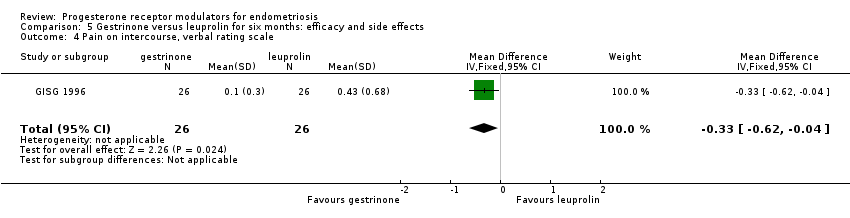
Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 4 Pain on intercourse, verbal rating scale.

Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 5 Non‐menstrual pain, visual analogue scale.

Comparison 5 Gestrinone versus leuprolin for six months: efficacy and side effects, Outcome 6 Side effects.

Comparison 6 Gestrinone 1.25 mg versus gestrinone 2.5 mg: efficacy and side effects, Outcome 1 improvement in pain.
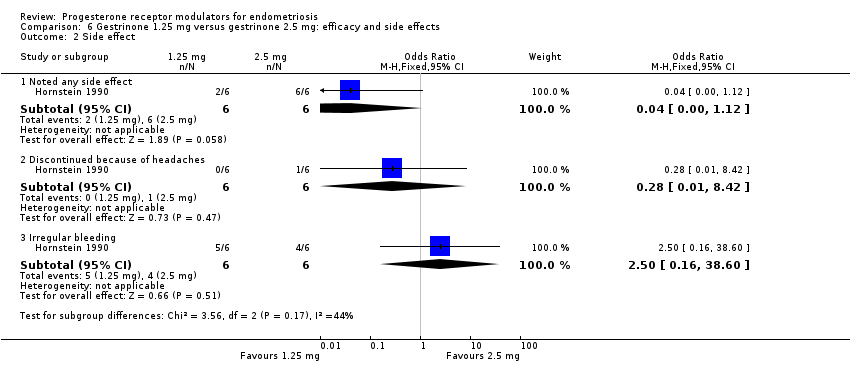
Comparison 6 Gestrinone 1.25 mg versus gestrinone 2.5 mg: efficacy and side effects, Outcome 2 Side effect.
| Mifepristone versus placebo for endometriosis | ||||||
| Patient or population: women with symptomatic endometriosis Settings: gynaecology clinic Intervention: progesterone receptor modulator (mifepristone) Comparison: placebo | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No. of participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Placebo | Mifepristone | |||||
| Prevalence of dysmenorrhoea Follow‐up: 3 months | 402 per 1000 | 51 per 1000 | OR 0.08 (0.04 to 0.17) | 352 | ⊕⊕⊕⊝ | |
| Prevalence of dyspareunia Follow‐up: 3 months | 288 per 1000 | 85 per 1000 | OR 0.23 (0.10 to 0.51) | 223 | ⊕⊕⊝⊝ | |
| Side effects: amenorrhoea Follow‐up: 3 months | 11 per 1000 | 884 per 1000 | OR 686.16 (92.29 to 5101.33) | 360 | ⊕⊕⊕⊝ High | 239/270 events in the mifepristone group vs 1/90 in the placebo group |
| Side effects: hot flushes Follow‐up: 3 months | 11 per 1000 | 243 per 1000 | OR 28.79 (3.93 to 210.73) | 360 | ⊕⊕⊕⊝ High | 66/270 events in the mifepristone group vs 1/90 in the placebo group |
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI) | ||||||
| GRADE Working Group grades of evidence | ||||||
| aDowngraded one level for serious imprecision (wide confidence intervals and/or very few events) bOutcome applied only to women with dysmenorrhoea at baseline, but this was 352/360 women randomised, so not downgraded for indirectness cOutcome applied only to women with dyspareunia at baseline, which was 223/360 women randomised. Downgraded one level for serious indirectness | ||||||
| Gestrinone versus danazol for endometriosis | ||||||
| Patient or population: women with symptomatic endometriosis Settings: gynaecology clinic Intervention: progesterone receptor modulator (gestrinone) Comparison: danazol | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No. of participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Danazol | Gestrinone | |||||
| Pelvic pain: none or mild | 890 per 1000 | 852 per 1000 (727 to 927) | OR 0.71 (0.33 to 1.56) | 230 (2) | ⊕⊕⊝⊝ | |
| Dysmenorrhoea: none or mild Follow‐up: 6 months | 721 per 1000 | 650 per 1000 | OR 0.72 (0.39 to 1.33) | 214 | ⊕⊝⊝⊝ | |
| Dyspareunia: none or mild Follow‐up: 6 months | 889 per 1000 | 869 per 1000 | OR 0.83 (0.37 to 1.86) | 222 | ⊕⊝⊝⊝ | |
| Side effects: hirsutism Follow‐up: 6 months | 248 per 1000 | 464 per 1000 | OR 2.63 (1.60 to 4.32) | 302 | ⊕⊝⊝⊝ | I2 = 68% |
| Decreased breast size Follow‐up: 6 months | 477 per 1000 | 360 per 1000 | OR 0.62 (0.38 to 0.98) | 302 | ⊕⊕⊝⊝ | |
| Side effects: hot flushes Follow‐up: 6 months | 425 per 1000 | 368 per 1000 (270 to 482) | OR 0.79 (0.50 to 1.26) | 302 (2 studies) | ⊕⊝⊝⊝ | I2 = 72% |
| Side effects: acne Follow‐up: 6 months | 556 per 1000 | 644 per 1000 (529 to 744) | OR 1.45 (0.90 to 2.33) | 302 (2 studies) | ⊕⊕⊝⊝ | |
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI) | ||||||
| GRADE Working Group grades of evidence | ||||||
| aAssessed in all randomised participants. Not all were symptomatic at baseline (although results show no significant differences in baseline symptoms between groups). Outcome therefore applies only to a select subgroup of participants: downgraded one level for serious indirectness bDowngraded one level for serious risk of bias associated with poor reporting of study methods, high attrition in one study, and high risk of other bias in both studies cImprecision of results (wide confidence intervals and/or few events), downgraded one level for serious imprecision dDowngraded one level for serious inconsistency | ||||||
| Gestrinone versus leuprolin for endometriosis | ||||||
| Patient or population: women with symptomatic endometriosis Settings: gynaecology clinic Intervention: progesterone receptor modulator (gestrinone) Comparison: leuprolin | ||||||
| Outcomes | Illustrative comparative risks* (95% CI) | Relative effect | No. of participants | Quality of the evidence | Comments | |
| Assumed risk | Corresponding risk | |||||
| Leuprolin | Gestrinone | |||||
| Dysmenorrhoea, verbal rating scale Follow‐up: 6 months | In control group, mean score for dysmenorrhoea on verbal rating scale was 0.04 points | Mean score in gestrinone group was 0.35 points higher (0.12 to 0.58 higher) | 55 | ⊕⊕⊝⊝ | Verbal rating scale defines dysmenorrhoea according to limitation of ability to work (mild = 1, moderate = 2, incapacitated = 3) | |
| Dyspareunia, verbal rating scale Follow‐up: 6 months | In control group, mean score for dyspareunia on verbal rating scale was 0.43 points | Mean score in gestrinone group was 0.33 points lower (0.62 to 0.04 lower) | 52 | ⊕⊕⊝⊝ | Verbal rating scale defines dyspareunia according to limitation of sexual activity (discomfort tolerated = 1; pain interrupts intercourse = 2, intercourse avoided owing to pain = 3) | |
| Amenorrhoea Follow‐up: 6 months | 962 per 1000 | 500 per 1000 | OR 0.04 (0.01 to 0.38) | 49 | ⊕⊕⊝⊝ | Only 55 events overall |
| Spotting or bleeding Follow‐up: 6 months | 38 per 1000 | 475 per 1000 | OR 22.92 (2.64 to 198.66) | 49 | ⊕⊕⊝⊝ | Only 12 events overall |
| Side effects: hot flushes Follow‐up: 6 months | 679 per 1000 | 297 per 1000 (112 to 571) | OR 0.20 (0.06 to 0.63) | 55 | ⊕⊕⊝⊝ | Only 27 events overall |
| *The basis for the assumed risk is the mean control group risk across studies. The corresponding risk (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). | ||||||
| GRADE Working Group grades of evidence | ||||||
| aDowngraded two levels for very serious imprecision: confidence intervals were compatible with no clinically meaningful difference between groups, or with small benefit in one group bDowngraded two levels for very serious imprecision: small overall sample size (n = 55) and low event rates | ||||||
| Outcome | Study | Comparison | Measure | Int group | Control group | P value | |
| Combined non‐pelvic pain, dysmenorrhoea, and dyspareunia | Asoprisnil: 5 mg (n = 31), 10 mg (n = 33), 25 mg (n = 32) Placebo (n = 34) | Mean reduction at 3 months on 0‐4 pain scale | 0.5 points for each dose | < 0.1 points | < 0.05 |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Dysmenorrhoea at 3 months Show forest plot | 1 | 352 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.08 [0.04, 0.17] |
| 1.1 Mifepristone 2.5 mg | 1 | 114 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.24 [0.09, 0.64] |
| 1.2 Mifepristone 5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.02 [0.00, 0.16] |
| 1.3 Mifepristone 10 mg | 1 | 118 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.02 [0.00, 0.17] |
| 2 Dyspareunia at 3 months Show forest plot | 1 | 223 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.23 [0.10, 0.51] |
| 2.1 Mifepristone 2.5 mg | 1 | 74 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.18, 2.13] |
| 2.2 Mifepristone 5 mg | 1 | 73 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.04 [0.00, 0.40] |
| 2.3 Mifepristone 10 mg | 1 | 76 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.13 [0.03, 0.60] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Amenorrhoea at 3 months Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 Mifepristone all doses | 1 | 360 | Odds Ratio (M‐H, Fixed, 95% CI) | 686.16 [92.29, 5101.33] |
| 2 Amenorrhoea at 3 months: subgroup analysis Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Mifepristone 2.5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 207.67 [27.50, 1568.36] |
| 2.2 Mifepristone 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 3916.0 [348.75, 43971.52] |
| 2.3 Mifepristone 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 3916.0 [348.75, 43971.52] |
| 3 Hot flushes at 3 months Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 3.1 Mifepristone all doses | 1 | 360 | Odds Ratio (M‐H, Fixed, 95% CI) | 28.79 [3.93, 210.73] |
| 4 Hot flushes at 3 months: subgroup analysis Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 Mifepristone 2.5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 19.24 [2.49, 148.54] |
| 4.2 Mifepristone 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 23.82 [3.11, 182.24] |
| 4.3 Mifepristone 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 46.76 [6.21, 351.92] |
| 5 Nausea at 3 months Show forest plot | 1 | 360 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.72 [0.20, 15.03] |
| 5.1 Mifepristone 2.5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 5.2 Mifepristone 5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.04, 25.76] |
| 5.3 Mifepristone 10 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.44 [0.12, 48.60] |
| 6 Vomiting at 3 months Show forest plot | 1 | 360 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.10, 10.01] |
| 6.1 Mifepristone 2.5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.04, 25.76] |
| 6.2 Mifepristone 5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 6.3 Mifepristone 10 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.02 [0.04, 25.76] |
| 7 Fatigue/Tiredness at 3 months Show forest plot | 1 | 360 | Odds Ratio (M‐H, Fixed, 95% CI) | 5.48 [0.71, 42.27] |
| 7.1 Mifepristone 2.5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.92 [0.21, 73.08] |
| 7.2 Mifepristone 5 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 7.3 Mifepristone 10 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 7.11 [0.40, 125.92] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Prevalence of dysmenorrhoea Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 1.1 2.5 mg vs 5 mg | 1 | 170 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.22, 3.29] |
| 1.2 5 mg vs 10 mg | 1 | 173 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.22 [0.32, 4.71] |
| 2 Dysmenorrhoea score 0‐10 VAS scale Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 5 mg vs 25 mg | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Prevalence of dyspareunia Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 3.1 2.5 mg vs 5 mg | 1 | 108 | Odds Ratio (M‐H, Fixed, 95% CI) | 6.37 [0.74, 54.81] |
| 3.2 5 mg vs 10 mg | 1 | 109 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.52 [0.05, 5.90] |
| 4 Dyspareunia score 0‐10 VAS scale Show forest plot | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 4.1 5 mg vs 25 mg | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Prevalence of pelvic pain Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 2.5 mg vs 5 mg | 1 | 110 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.81 [0.63, 5.17] |
| 5.2 5 mg vs 10 mg | 1 | 120 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.97 [0.79, 19.97] |
| 6 Prevalence of amenorrhoea Show forest plot | 2 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 6.1 2.5 mg vs 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.45 [0.21, 0.97] |
| 6.2 5 mg vs 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.10 [0.47, 2.56] |
| 6.3 5 mg vs 25 mg | 1 | 26 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.61 [0.08, 4.41] |
| 7 Prevalence of hot flushes Show forest plot | 2 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 7.1 2.5 mg vs 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.38, 1.89] |
| 7.2 5 mg vs 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.75 [0.35, 1.58] |
| 7.3 5 mg vs 25 mg | 1 | 26 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.24 [0.12, 87.13] |
| 8 Prevalence of nausea Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 8.1 2.5 mg vs 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.02 [0.18, 22.71] |
| 8.2 5 mg vs 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.06, 16.24] |
| 9 Prevalence of vomiting Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 9.1 2.5 mg vs 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.03 [0.12, 75.46] |
| 9.2 5 mg vs 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.20] |
| 10 Prevalence of fatigue/tiredness Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 10.1 2.5 mg vs 5 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 13.92 [0.77, 250.94] |
| 10.2 5 mg vs 10 mg | 1 | 180 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.03 [0.00, 0.54] |
| 11 Prevalence of endometrial thickness > 20 mm Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 11.1 5 mg vs 25 mg | 1 | 26 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.41 [0.28, 7.13] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 None or mild pelvic pain Show forest plot | 2 | 230 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.33, 1.56] |
| 2 None or mild dysmenorrhoea Show forest plot | 2 | 214 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.72 [0.39, 1.33] |
| 3 None or mild dyspareunia Show forest plot | 2 | 222 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.37, 1.86] |
| 4 Adverse effects Show forest plot | 2 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 4.1 Acne | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.45 [0.90, 2.33] |
| 4.2 Seborrhoea | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.73 [1.67, 4.46] |
| 4.3 Hirsutism | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.63 [1.60, 4.32] |
| 4.4 Voice problems | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.70 [0.34, 1.43] |
| 4.5 Swelling hands/feet | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.40 [0.82, 2.38] |
| 4.6 Hot flushes | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.79 [0.50, 1.26] |
| 4.7 Decreased breast size | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.38, 0.98] |
| 4.8 Leg or muscle cramps | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.49 [0.30, 0.78] |
| 4.9 Headache | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.36 [0.84, 2.21] |
| 4.10 Nausea | 2 | 302 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.36 [0.84, 2.19] |
| 4.11 Vomiting | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.32, 1.43] |
| 4.12 Loss of appetite | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.31 [0.72, 2.37] |
| 4.13 Hunger | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.59 [0.36, 0.97] |
| 4.14 Dizziness | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.24 [0.75, 2.05] |
| 4.15 Tiredness | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.44 [0.84, 2.45] |
| 4.16 Faintness | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.23 [0.54, 2.76] |
| 4.17 Skin rash | 1 | 264 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.71 [0.91, 3.20] |
| 4.18 Weight gain | 1 | 38 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.34 [0.09, 1.27] |
| 4.19 Vaginal dryness | 1 | 38 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.18 [0.01, 4.00] |
| 4.20 Raised liver transaminases | 1 | 38 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.18 [0.01, 4.00] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 Painful periods, visual analogue scale Show forest plot | 1 | 55 | Mean Difference (IV, Fixed, 95% CI) | 0.82 [0.15, 1.49] |
| 2 Painful periods, verbal rating scale Show forest plot | 1 | 55 | Mean Difference (IV, Fixed, 95% CI) | 0.35 [0.12, 0.58] |
| 3 Pain on intercourse, visual analogue scale Show forest plot | 1 | 52 | Mean Difference (IV, Fixed, 95% CI) | ‐1.16 [‐2.08, ‐0.24] |
| 4 Pain on intercourse, verbal rating scale Show forest plot | 1 | 52 | Mean Difference (IV, Fixed, 95% CI) | ‐0.33 [‐0.62, ‐0.04] |
| 5 Non‐menstrual pain, visual analogue scale Show forest plot | 1 | 55 | Mean Difference (IV, Fixed, 95% CI) | ‐0.41 [‐1.76, 0.94] |
| 6 Side effects Show forest plot | 1 | 813 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.42, 1.01] |
| 6.1 Seborrhoea | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.23 [0.13, 82.71] |
| 6.2 Swelling hands/feet | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 5.59 [0.26, 121.96] |
| 6.3 Hot flushes | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.20 [0.06, 0.63] |
| 6.4 Leg or muscle cramps | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.55] |
| 6.5 Headache | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.20 [0.06, 0.63] |
| 6.6 Nausea | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.06, 17.49] |
| 6.7 Dizziness | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.16 [0.18, 25.32] |
| 6.8 Skin rash | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 3.23 [0.13, 82.71] |
| 6.9 Vaginal dryness | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.19 [0.01, 4.21] |
| 6.10 Mood changes | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.10, 4.34] |
| 6.11 Joint pain | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.16 [0.18, 25.32] |
| 6.12 Drowsiness | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.16 [0.18, 25.32] |
| 6.13 Tachycardia | 1 | 55 | Odds Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.06, 17.49] |
| 6.14 Amenorrhoea | 1 | 49 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.04 [0.01, 0.38] |
| 6.15 Spotting or bleeding | 1 | 49 | Odds Ratio (M‐H, Fixed, 95% CI) | 22.92 [2.64, 198.66] |
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
| 1 improvement in pain Show forest plot | 1 | 10 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.27 [0.01, 8.46] |
| 2 Side effect Show forest plot | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 2.1 Noted any side effect | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.04 [0.00, 1.12] |
| 2.2 Discontinued because of headaches | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.28 [0.01, 8.42] |
| 2.3 Irregular bleeding | 1 | 12 | Odds Ratio (M‐H, Fixed, 95% CI) | 2.5 [0.16, 38.60] |


