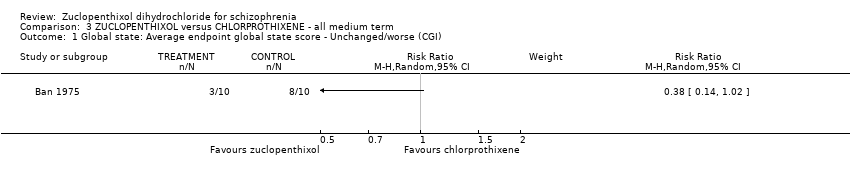
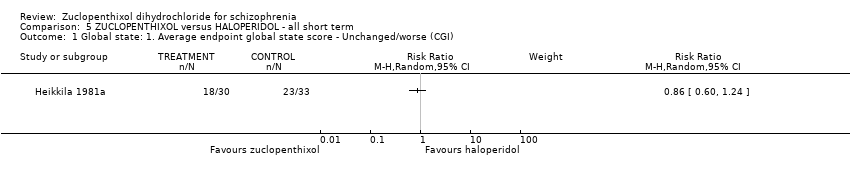
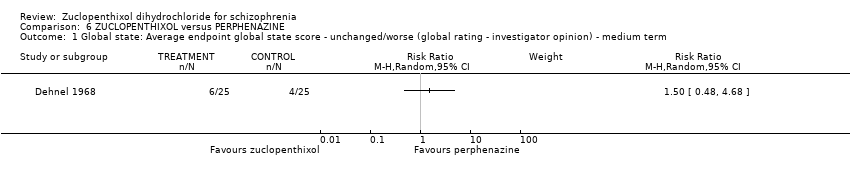
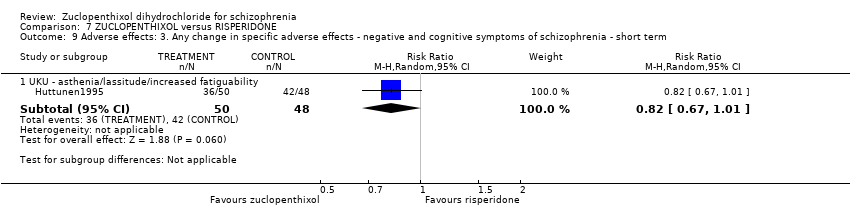
| 1 Global state: 1. Average endpoint global state score ‐ Unchanged/worse (CGI, scores not reported) Show forest plot | 2 | 135 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.75, 1.13] |
|
| 2 Global state: 2. Average endpoint global state score ‐ No Recovery Show forest plot | 1 | 64 | Risk Ratio (M‐H, Random, 95% CI) | 1.02 [0.89, 1.16] |
|
| 3 Global state: 3a. Average endpoint global state score (GAS, high score not reported, average score = 63.4) Show forest plot | 1 | 60 | Mean Difference (IV, Random, 95% CI) | ‐0.60 [‐8.12, 6.92] |
|
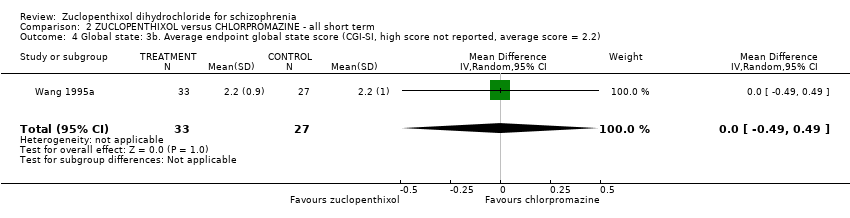
| 4 Global state: 3b. Average endpoint global state score (CGI‐SI, high score not reported, average score = 2.2) Show forest plot | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 0.0 [‐0.49, 0.49] |
|
| 5 Mental state: 1. No clinically important change in general mental state ‐ Not improved (PANSS, scores not reported) Show forest plot | 1 | 120 | Risk Ratio (M‐H, Random, 95% CI) | 0.98 [0.81, 1.18] |
|
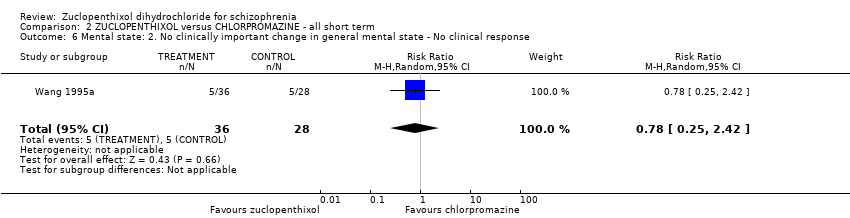
| 6 Mental state: 2. No clinically important change in general mental state ‐ No clinical response Show forest plot | 1 | 64 | Risk Ratio (M‐H, Random, 95% CI) | 0.78 [0.25, 2.42] |
|
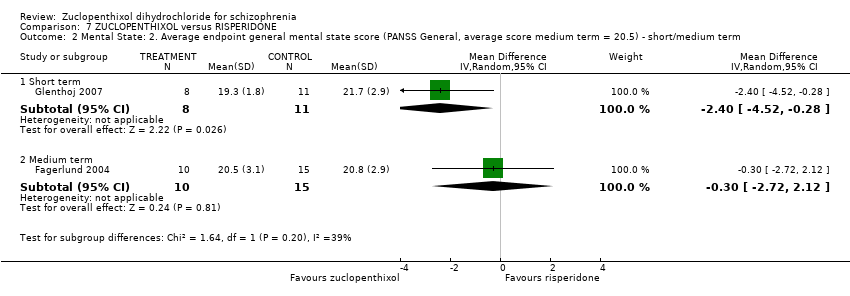
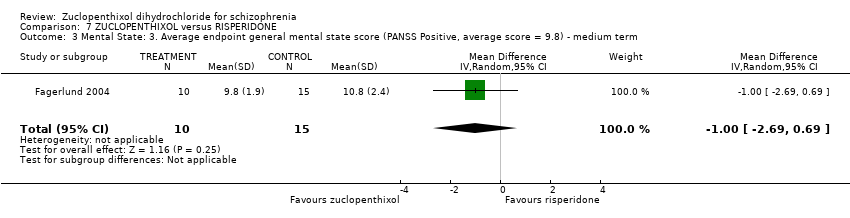
| 7 Mental state: 3. Average endpoint general mental state score (BPRS, high score = 34.2) Show forest plot | 3 | 221 | Mean Difference (IV, Random, 95% CI) | 0.40 [‐2.43, 3.23] |
|
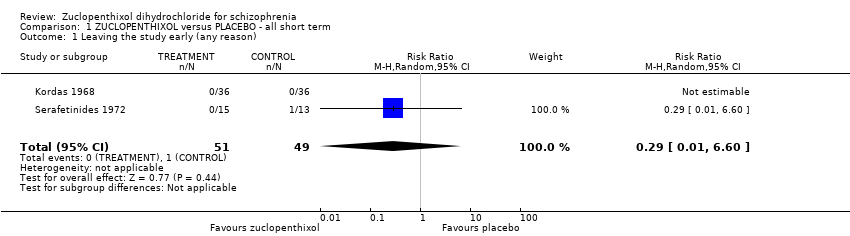
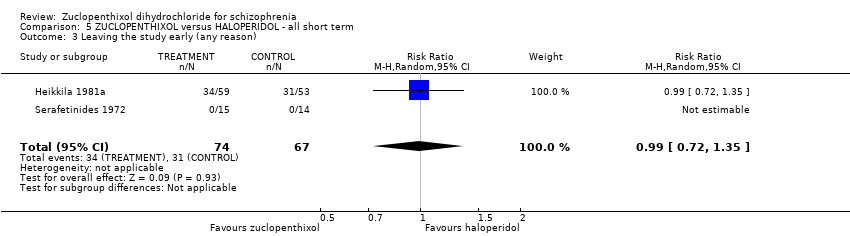
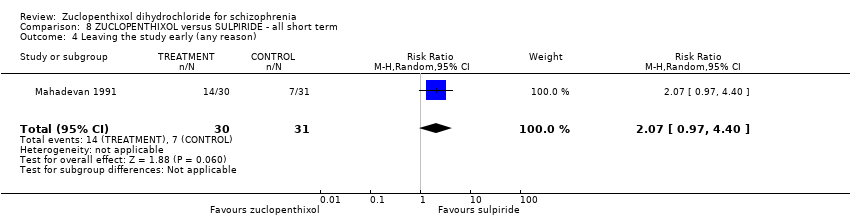
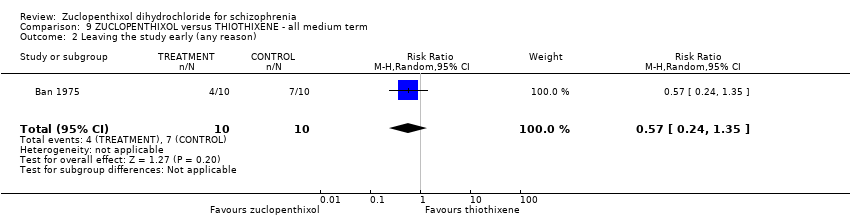
| 8 Leaving the study early (any reason) Show forest plot | 6 | 766 | Risk Ratio (M‐H, Random, 95% CI) | 0.54 [0.36, 0.81] |
|
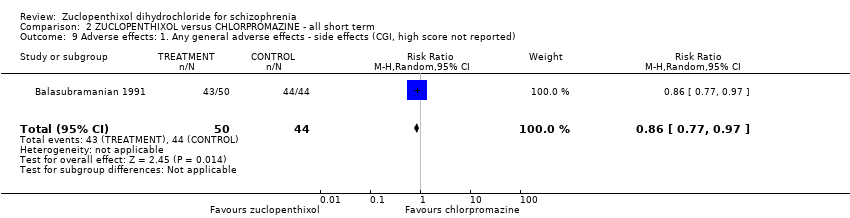
| 9 Adverse effects: 1. Any general adverse effects ‐ side effects (CGI, high score not reported) Show forest plot | 1 | 94 | Risk Ratio (M‐H, Random, 95% CI) | 0.86 [0.77, 0.97] |
|
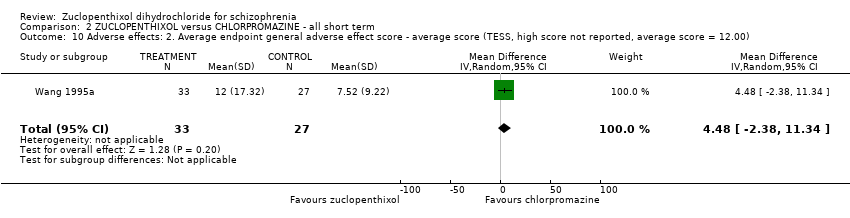
| 10 Adverse effects: 2. Average endpoint general adverse effect score ‐ average score (TESS, high score not reported, average score = 12.00) Show forest plot | 1 | 60 | Mean Difference (IV, Random, 95% CI) | 4.48 [‐2.38, 11.34] |
|
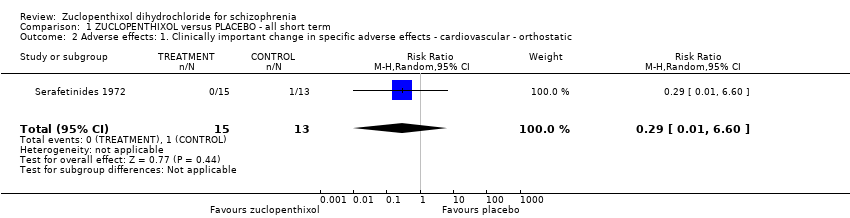
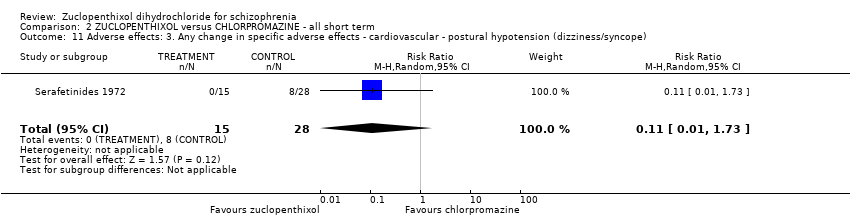
| 11 Adverse effects: 3. Any change in specific adverse effects ‐ cardiovascular ‐ postural hypotension (dizziness/syncope) Show forest plot | 1 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.01, 1.73] |
|
| 12 Adverse effects: 4. Any change in specific adverse effects ‐ central nervous system ‐ arousal Show forest plot | 2 | | Risk Ratio (M‐H, Random, 95% CI) | Subtotals only |
|
| 12.1 excitation | 1 | 43 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.07, 5.47] |
| 12.2 sedation | 2 | 163 | Risk Ratio (M‐H, Random, 95% CI) | 1.11 [0.73, 1.70] |
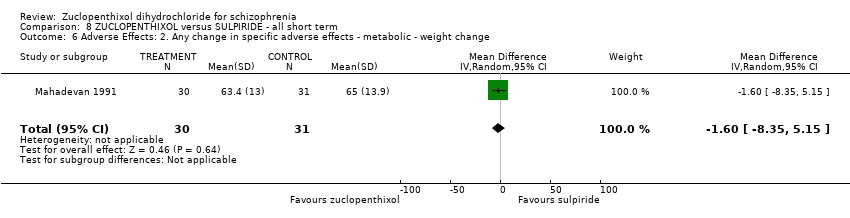
| 13 Adverse effects: 5. Any change in specific adverse effects ‐ metabolic ‐ weight change ‐ loss or gain of weight of 10 pounds Show forest plot | 1 | 29 | Risk Ratio (M‐H, Random, 95% CI) | 0.62 [0.22, 1.75] |
|
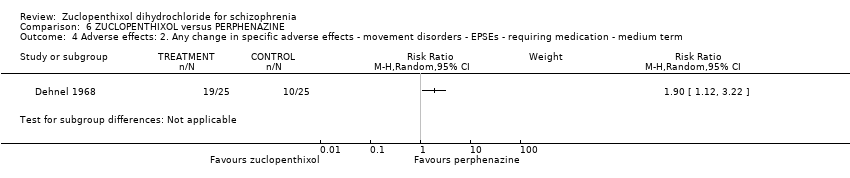
| 14 Adverse effects: 6a. Any change in specific adverse effects ‐ movement disorders ‐ EPSEs Show forest plot | 3 | 199 | Risk Ratio (M‐H, Random, 95% CI) | 0.94 [0.61, 1.45] |
|
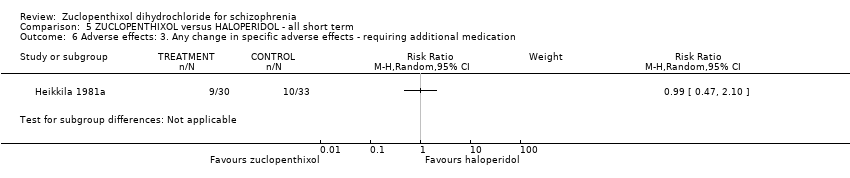
| 15 Adverse effects: 6b. Any change in specific adverse effects ‐ movement disorders ‐ additional medication use Show forest plot | | | Other data | No numeric data |
|